
Concept explainers
Interpretation:Primary, secondary and tertiary hydrogen in structures of the isohexaneshould be labeled.
Concept introduction: In accordance with IUPAC convention, the longest chain can be found from either direction provided it is longest and digits indicate the position of carbon or the position of branched alkyl chain in cases of branched hydrocarbons. All the side chains are named in alphabetical order.
The IUPAC system for nomenclature of straight hydrocarbon makes use of table given as follows:
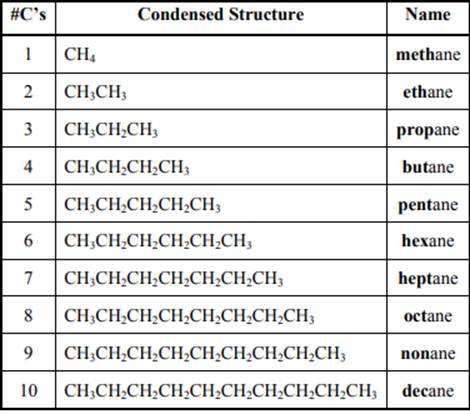
Beside the IUPAC names there are certain common names. The common prefixes include prefixes“iso-” and “neo-”.For example,isobutane is common name used popularly for
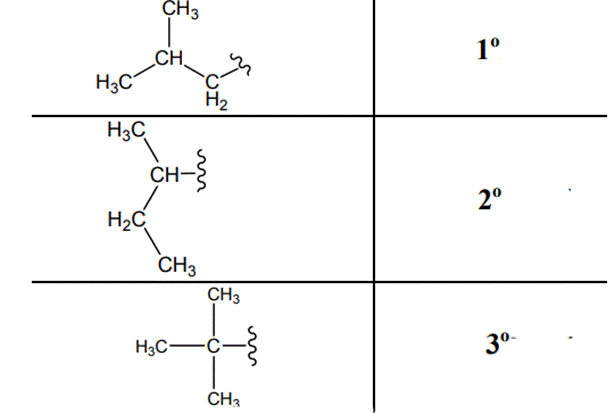
The carbon linked to one alkyl/carbon while other two
The carbon linked to two alkyl /carbons and one
The carbon linked to three alkyl groups/carbons and no
These are indicated as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Organic Chemistry: Structure and Function
- Choose the correct answer. Choose... Choose.. An aldehyde with molecular formula C2H6O A ketone with molecular formula C3H60 A ketone with molecular formula C2H60 An alkene with molecular formula C5H10 An alcohol with molecular formula C4H8O A saturated hydrocarbon with molecular formula C6H14 An unsaturated hydrocarbon with molecular formula C6H12 An alkane with molecular formula C5H12 Choose...arrow_forwardHere is the chemical structure of 2-bromobutane: H H. H C-C-H H :Br: H H Decide whether each molecule in the table below is another molecule of 2-bromobutane, a molecule of an isomer of 2-bromobutane, or a molecule of an entirely different compound. molecule relationship to 2-bromobutane CH, (Choose one) H,c=ċ=CH,-Br Explanation Check O 2021 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center I Accessibility P Type here to search 76°F DELL F1 F2 F3 F4 F5 FB F9 F10 F11 F12 PrtScr PAA @ %23 2$ 1 8. 9 Q W R Y. A F G H. K V N4 Alt Ctri Alt Σ B. C1arrow_forwardWrite the systematic name of each organic molecule: CH3 CH3 O || || structure CH- CH3 OH | CH-CH₂- OH CH₂ CH₂-OH || CH3 CH₂ - CH-C-CH3 CH3 name 0arrow_forward
- Write the systematic name of each organic molecule: O || CH3–CH2–CH2–CH2−C−N–CH2–CH2–CH3 H₂N CH3 CH₂- structure || || CH₂ CH₂ CH3 C-CH₂-CH3 NH C−NH–CH,—CH,—CH3 name 1 0arrow_forwardWrite the systematic name of each organic molecule: structure O H-C-CH-CH3 OH || CH3–CH2–CH2–CH-C-H CH3 O OH 0=0 HO–CH2–CH2–CH2CH-C-H name 0 | 0arrow_forwardA student named the molecule shown below as 5-heptene. Explain the error made by the student and provide the correct name. H H H H H-C C. -C H -H HH C C—C—C—H H HHH Harrow_forward
- Worksheet 1: IUPAC Nomenclature Use the IUPAC Nomenclature to correctly name the following organic molecules. Name Name H H H H H H FC C C 4 H2C- -CH3 H H. H. H Name Name ннн нн H H H 1 C 21 3 4 C C -H H-C C=C-C-C--H CH3 H H нн Name Name H CH3 H. H Н—С — Н H H H- с —Н CH3 H. H. H H Name Name C2H5 H H H нннн нн 1 2 3 4 6. H- -H H-C-C-CFC,-C-C-H H H H CH3 H. H H H Н-С-н H.arrow_forwardLand Nav.zip Name the following molecule: H H H H HH H H HMCH HH-C-HH C H C-H H H Give IUPAC names for the following compounds: H³C CH3 H₂C=CHCHCCH³ BIOL 304 Lab 12 (...docx) 1 CH3 CH3 CH3CH₂CH=CCH₂CH3 A H BIOL 304 Lab 11 (....docx) IMG-5847 (1).jpg Pagearrow_forwardWrite the systematic name of each organic molecule: CH3 structure OH OH | | CH-CH-CH - CH3 OH OH | HỌ—CH2–CH–CH2–CH2 -CH₂ OH OH CH3 - CH₂ CH-CH₂CH3 name 0 0 X 5arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
