
Organic Chemistry: Structure and Function
8th Edition
ISBN: 9781319079451
Author: K. Peter C. Vollhardt, Neil E. Schore
Publisher: W. H. Freeman
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 2, Problem 42P
Interpretation Introduction
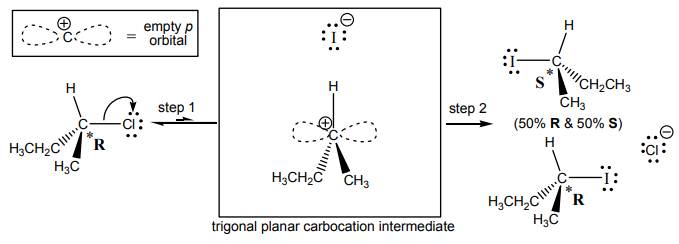
Interpretation: Three discrete steps shown for conversion of alcohol to
Concept introduction: Alcohol to haloalkane conversion is essentially unimolecular nucleophilic substitution. These substitutions proceed via carbocation formation that occurs in a slow step; followed by a rapid step that involves attack of nucleophile to carbocation center. The products obtained are racemic in the sense that each of the two enantiomers is formed in equal amounts. The general pathway of such substitution is illustrated as follows:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw what would happen to 2,5-dimethyl-2-hexene in an alkyl halides and alkane reaction. This is for a concept map. So what would the alkene 2,5-dimethyl-2-hexene be as an alkyl halides and how to you go from an alkene to an alkane with 2,5-dimethyl-2-hexene. No bond lines. I know this much for alkene to alkane
This is an organic chemistry question?
Identify the major product(s) formed by the following reaction.
PLEASE SHOW STEP BY STEP! ALSO PLEASE INCLUDE ALL ELECTRON LONE PAIRS AND CHARGES FOR STRUCTURES IF NECESSARY!
Kindly answer the following questions from 1 to 3. As per your guidelines, only solve the first three questions.
1. Draw the Lewis structures and predict the hybridization (sp, sp2, sp3), geometry (linear, trigonal, tetrahedral) and bond angles (180°, 120°, 109.5°) for the central atoms in the following compounds.
2. Give the systematic (IUPAC) name for each of the following alkanes, cycloalkanes, bicycloalkanes and alkyl halides.
3. Given the IUPAC name, draw the structure of the following compounds.
Chapter 2 Solutions
Organic Chemistry: Structure and Function
Ch. 2.1 - Prob. 2.1ECh. 2.1 - Prob. 2.2ECh. 2.1 - Prob. 2.3ECh. 2.1 - Prob. 2.5TIYCh. 2.1 - Prob. 2.6ECh. 2.1 - Prob. 2.7ECh. 2.2 - Prob. 2.8ECh. 2.3 - Prob. 2.9ECh. 2.3 - Prob. 2.10ECh. 2.3 - Prob. 2.11E
Ch. 2.3 - Prob. 2.12ECh. 2.3 - Prob. 2.13ECh. 2.3 - Prob. 2.14ECh. 2.3 - Prob. 2.15ECh. 2.3 - Prob. 2.17TIYCh. 2.3 - Prob. 2.19TIYCh. 2.5 - Prob. 2.20ECh. 2.6 - Prob. 2.21ECh. 2.6 - Prob. 2.22ECh. 2.6 - Prob. 2.23ECh. 2.6 - Prob. 2.25TIYCh. 2.7 - Prob. 2.26ECh. 2.9 - Prob. 2.28TIYCh. 2 - Prob. 31PCh. 2 - Prob. 32PCh. 2 - Prob. 33PCh. 2 - Prob. 34PCh. 2 - Prob. 35PCh. 2 - Prob. 36PCh. 2 - Prob. 37PCh. 2 - Prob. 38PCh. 2 - Prob. 39PCh. 2 - Prob. 40PCh. 2 - Prob. 41PCh. 2 - Prob. 42PCh. 2 - Prob. 43PCh. 2 - Prob. 44PCh. 2 - Prob. 45PCh. 2 - Prob. 46PCh. 2 - Prob. 47PCh. 2 - Prob. 48PCh. 2 - Prob. 49PCh. 2 - Prob. 50PCh. 2 - Prob. 51PCh. 2 - Prob. 52PCh. 2 - Prob. 53PCh. 2 - Prob. 54PCh. 2 - Prob. 55PCh. 2 - Prob. 56PCh. 2 - Prob. 57PCh. 2 - Prob. 58PCh. 2 - Prob. 59PCh. 2 - Prob. 60PCh. 2 - Prob. 61PCh. 2 - Prob. 62PCh. 2 - Prob. 63PCh. 2 - Prob. 64PCh. 2 - Prob. 65PCh. 2 - Prob. 66P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Identify (circle and name) all the functional groups in the following structures of the reactant and product. There may be functional groups that were not discussed in the video, so please use the functional groups reference sheet to assist you.arrow_forward7. Draw curved arrows indicating the movements of electrons between the following pair of resonance structures. Name the pattern of resonance shown. What is the hybridization of the carbon atoms? 8. Draw the remaining three resonance structures for the molecule in problem 7 above. 9. There are several possible forms of a trisubstituted cyclohexane with the formula C10H200. I have drawn four of them. From these, which one do you think is most commonly naturally occurring, and why? Which is least commonly occurring and why? HO HO" HO HOarrow_forwardHint: In naming ketones, the -e ending of the parent alkane is replaced with the -one suffix of the ketone family. The longest carbon chain is numbered to give the carbonyl carbon the lowest possible number.arrow_forward
- Next, let's explore what happens when you remove two hydrogens from the structure above and form ethene, CH,CH,. How many bonds should be between the two carbons to give each carbon a total of four bonds? Draw the structural formula for ethene, CH,CH,.arrow_forwardcomplete the two tables attached below (alkyl halide is 4 seperate structures so there are four different answers provide me with all four of those)arrow_forwardPlease answer all of it. If not, please skip. Draw the structure of a branched hydrocarbon that also contains at least one atom with trigonal planar geometry. Draw the structure of an aromatic hydrocarbon containing two halogens. Draw the structure of a molecule containing an aliphatic primary amine and a carboxyl group. Draw the structure of an acyclic molecule containing a primary and a secondary alcohol group. Draw the structure of an achiral tertiary alcoholarrow_forward
- 3) Starting from any alkene, show how you could arrive at molecular entities containing the corresponding functional groups aldehyde, alkyne or ester.arrow_forwardDescribe (meaning what you would do and observe) simple (not including melting points, boiling points, cleaving reactions, or instrumental analysis) chemical tests (if any) that would distinguish between the compounds named below. [Hing: using a table may be helpful] • 1-hexyne • cyclopentene • 2-methylbutane • 2-methyl-2-propanolarrow_forwardQuestion: In an organic chemistry laboratory, a student is performing a reaction to synthesize a compound. The reaction involves the conversion of an alcohol into an alkene using a dehydration process. The student chooses to use concentrated sulfuric acid (H2SO4) as the catalyst for this reaction. However, during the reaction, the student notices that another product is formed along with the desired alkene. (a) What is the likely identity of the unexpected product formed during the reaction? Explain your answer. (b) Provide a plausible explanation for the formation of the unexpected product and suggest a modification to the reaction conditions that would favor the desired alkene as the major product.arrow_forward
- 6. Which of the following is an ester with molecular formula C6H10O₂? (If more than one answer is correct circle all correct answers). x 0- A 0 B To X و میری D x 7. Indicate the orbitals that are overlapping to give each of the indicated bonds (for example Csp3- Hs). Keep in mind that not all hydrogens or lone pairs are drawn in. C5P3 E Xarrow_forward2. Consider the following organic molecules A-H. Many of them are natural products isolated from Nature. Please answer the following questions by writing the Letter pertaining to each molecule next to the question. -N- H;C "CO,H N. Но CH2 ČH3 B strychnine (a plant alkaloid) hirsutic acid A nepetalactone (essence of cutnip) (a fimgul metabolito) H- H. I CH,0 E helminthosporal (a imgal toxu) D progesterone F cantharidin (a progestin hommone) (am insect vesicant) Br NO2 Br CI G untenine C (E)-1,6-dibromo-2,7-dichloro-3,7-dimethyloct-3-ene (fom red algae) (4 sponge metabolite)arrow_forwardOrganic Chemistry HW: CANNOT BE HAND DRAWN 2,6-dimethyloct-2-ene Hydrogen Bonding with Water use a computer program and illustrate the expanded structure of your molecule. (If the structure already contains any dashed lines, make them solid (non-dashed) covalent bonds for this assignment so that they are not confused with hydrogen bonds) Illustrate all the locations where your molecule could form hydrogen bonds with water either as a hydrogen donor or as a target (receiver) of hydrogen bonds from water. Do this by drawing bent water molecules as necessary and representing hydrogen bonds between water and the molecule using dashed lines. Be sure it is clear exactly which atoms on each molecule are involved in the hydrogen bonds. If your molecule is not capable of forming hydrogen bonds with water explain why not.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning