
Organic Chemistry: Structure and Function
8th Edition
ISBN: 9781319079451
Author: K. Peter C. Vollhardt, Neil E. Schore
Publisher: W. H. Freeman
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 2, Problem 64P
Interpretation Introduction
Interpretation: The
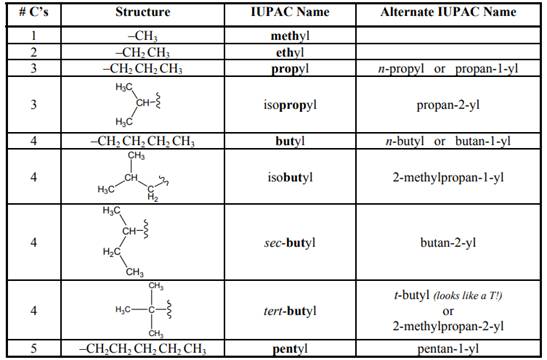
Concept introduction:As per IUPAC recommendations longest chain found in a continuous manner in a branched molecule is chosen as parent chain.
The IUPAC name begins with prefix to designate the number of
Names of branches that occur commonly as side chains are listed below:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify the best reagents to complete
the following reaction.
CI
N.
1. Name the following:
0
|||
a.
b.
C.
0
i
e.
CH
N
CH₂CH3
CH
d. CH3CH₂
CI
CH,COOH
C.
CH₂C=N
CH
CH3 CH₂CH₂CH3
OCH₂CH₂CH3
13)-Google Chrome
od/quiz/attempt.php?attempt%315766088cmid%=813838&page312#question-1763030-41
- (Academic)
Use one of the terms SNI, SN2, El or E2 to describe each of the following chemical reactions
Choose the correct match for each
NaNH,
Choose...
OTS
NH3
CH;CH,O-Na+,
Choose.. +
ethanol
КОН
Но
Br
DMSO
Choose...
Chapter 2 Solutions
Organic Chemistry: Structure and Function
Ch. 2.1 - Prob. 2.1ECh. 2.1 - Prob. 2.2ECh. 2.1 - Prob. 2.3ECh. 2.1 - Prob. 2.5TIYCh. 2.1 - Prob. 2.6ECh. 2.1 - Prob. 2.7ECh. 2.2 - Prob. 2.8ECh. 2.3 - Prob. 2.9ECh. 2.3 - Prob. 2.10ECh. 2.3 - Prob. 2.11E
Ch. 2.3 - Prob. 2.12ECh. 2.3 - Prob. 2.13ECh. 2.3 - Prob. 2.14ECh. 2.3 - Prob. 2.15ECh. 2.3 - Prob. 2.17TIYCh. 2.3 - Prob. 2.19TIYCh. 2.5 - Prob. 2.20ECh. 2.6 - Prob. 2.21ECh. 2.6 - Prob. 2.22ECh. 2.6 - Prob. 2.23ECh. 2.6 - Prob. 2.25TIYCh. 2.7 - Prob. 2.26ECh. 2.9 - Prob. 2.28TIYCh. 2 - Prob. 31PCh. 2 - Prob. 32PCh. 2 - Prob. 33PCh. 2 - Prob. 34PCh. 2 - Prob. 35PCh. 2 - Prob. 36PCh. 2 - Prob. 37PCh. 2 - Prob. 38PCh. 2 - Prob. 39PCh. 2 - Prob. 40PCh. 2 - Prob. 41PCh. 2 - Prob. 42PCh. 2 - Prob. 43PCh. 2 - Prob. 44PCh. 2 - Prob. 45PCh. 2 - Prob. 46PCh. 2 - Prob. 47PCh. 2 - Prob. 48PCh. 2 - Prob. 49PCh. 2 - Prob. 50PCh. 2 - Prob. 51PCh. 2 - Prob. 52PCh. 2 - Prob. 53PCh. 2 - Prob. 54PCh. 2 - Prob. 55PCh. 2 - Prob. 56PCh. 2 - Prob. 57PCh. 2 - Prob. 58PCh. 2 - Prob. 59PCh. 2 - Prob. 60PCh. 2 - Prob. 61PCh. 2 - Prob. 62PCh. 2 - Prob. 63PCh. 2 - Prob. 64PCh. 2 - Prob. 65PCh. 2 - Prob. 66P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 40- Bond angle in acetylene is. A - 120⁰ B - 180⁰ C-109⁰ A B 41- Bond angle in acetylene is. A-120⁰ B-1800 C - 109⁰ Barrow_forward1. CH, CH, - CH,-CH,-CH, + HO 2. H₂C-CH=CH₂ + HBr/H₂O₂ 3. H₂C-CH (OH)-CH₂ + H* 4. Nitrobenzene +? ? anilinearrow_forwardCONH₂ CH3 CH3 H₂C=CHCHCH₂CO₂CH3 1. LiAlH4 2. H₂O 1. LiAlH4 2. H3O+arrow_forward
- The general formal for alkanes - 3 .compounds is A - CnH2n. O B - CnHn+2. O C - CnH2n+2. O General formula of aromatic - 4 .compounds are A - CnH2n. O B - CnHn+2. O C - CnH2n-2. Oarrow_forward4. Draw the following compounds a. 1-chloro-4-ethyl-3-methylnonane [ Gatusofom gnit b. 4-methyl-2-pentyne Job i anticas) enod totall xbacd bigin i emoczopertori c. 1-bromo3-methylbutane d. 4-ethyl-3-fluoro-1-hexenearrow_forward2. Lone pair occupies a hybridized p-orbital allowing it to delocalization to a nearby side-way overlap. a. True b. Falsearrow_forward
- H2N -CH-c-OH 4. CH3 Neutral/acidic/ basic Polar/ non-polar Hydrophobic/hydrophilic. H2- N -сн—с—он CH2 CH2 O: 5. NH2 Neutral/acidic/ basic Polar/ non-polar Hydrophobic/hydrophilic.arrow_forwardb. CH3 CH3- CH3 CH3- a. H OH «НИ H OH CH3- CH3 CH3 CH3- O: Ho Ho Hooarrow_forwardan alkyne 1 eq. Br₂ HgSO H₂SO4. H₂O 1) Na. NH₂(1) 2) Br₂ Br +arrow_forward
- elsitini bos wei0 (8 5) Draw and build ethene, CH¿CH2. Be sure your drawing correctly reflects the geometry in the model.arrow_forward1, 3, 2 d. 3, 1, 2 e. 3, 2, 1 Choose the correct specific configuration (R & S) for C2 and C3 in the Question 25 structure (pdf document). a. C2 (R) & C3 (S) b. C2 (S) & C3 (S) c. C2 (S) & C3 (R) d. C2 (R) & C3 (R) US PAGE NEXT gationarrow_forwardPlease do the following question. Step by step A. 1-butyne + HgSO4, H2SO4 and H2O B. 2-methyl-1,3-butadiene + HC ≡ C – C ≡ Narrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY