
Organic Chemistry: Structure and Function
8th Edition
ISBN: 9781319079451
Author: K. Peter C. Vollhardt, Neil E. Schore
Publisher: W. H. Freeman
expand_more
expand_more
format_list_bulleted
Question
Chapter 2, Problem 62P
Interpretation Introduction
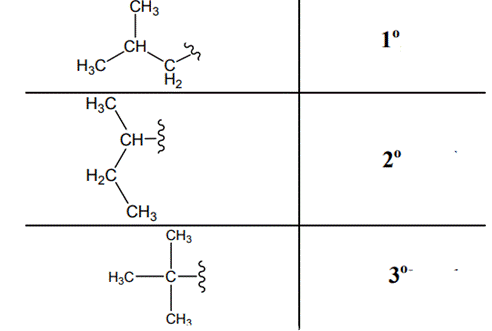
Interpretation: Primary, secondary and tertiary hydrogen in structure of
Concept introduction: The carbon linked to one alkyl / carbon while other two
The carbon linked to two alkyl /carbons and one
The different kinds of primary, secondary and tertiary hydrogen are illustrated as follows:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Cl
Br
h.
-CH3
Br
T
H₂C-CH
i.
CH
Cl
CH₂CH3
2.Label the following as R or S
H
a.
b.
C.
H
NH2
Brww
H
d. HO
O H₂N H
HO
B.
CH₂OH
c=0
HO-C-H
CH₂OH
+ CuSO
Chapter 2 Solutions
Organic Chemistry: Structure and Function
Ch. 2.1 - Prob. 2.1ECh. 2.1 - Prob. 2.2ECh. 2.1 - Prob. 2.3ECh. 2.1 - Prob. 2.5TIYCh. 2.1 - Prob. 2.6ECh. 2.1 - Prob. 2.7ECh. 2.2 - Prob. 2.8ECh. 2.3 - Prob. 2.9ECh. 2.3 - Prob. 2.10ECh. 2.3 - Prob. 2.11E
Ch. 2.3 - Prob. 2.12ECh. 2.3 - Prob. 2.13ECh. 2.3 - Prob. 2.14ECh. 2.3 - Prob. 2.15ECh. 2.3 - Prob. 2.17TIYCh. 2.3 - Prob. 2.19TIYCh. 2.5 - Prob. 2.20ECh. 2.6 - Prob. 2.21ECh. 2.6 - Prob. 2.22ECh. 2.6 - Prob. 2.23ECh. 2.6 - Prob. 2.25TIYCh. 2.7 - Prob. 2.26ECh. 2.9 - Prob. 2.28TIYCh. 2 - Prob. 31PCh. 2 - Prob. 32PCh. 2 - Prob. 33PCh. 2 - Prob. 34PCh. 2 - Prob. 35PCh. 2 - Prob. 36PCh. 2 - Prob. 37PCh. 2 - Prob. 38PCh. 2 - Prob. 39PCh. 2 - Prob. 40PCh. 2 - Prob. 41PCh. 2 - Prob. 42PCh. 2 - Prob. 43PCh. 2 - Prob. 44PCh. 2 - Prob. 45PCh. 2 - Prob. 46PCh. 2 - Prob. 47PCh. 2 - Prob. 48PCh. 2 - Prob. 49PCh. 2 - Prob. 50PCh. 2 - Prob. 51PCh. 2 - Prob. 52PCh. 2 - Prob. 53PCh. 2 - Prob. 54PCh. 2 - Prob. 55PCh. 2 - Prob. 56PCh. 2 - Prob. 57PCh. 2 - Prob. 58PCh. 2 - Prob. 59PCh. 2 - Prob. 60PCh. 2 - Prob. 61PCh. 2 - Prob. 62PCh. 2 - Prob. 63PCh. 2 - Prob. 64PCh. 2 - Prob. 65PCh. 2 - Prob. 66P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Br CH3 a. Common Name OH NO 2 Br b. Common Name || H3C-CH-CH-CH2 C- -NH-CH2-CH3 С. IUPAC || H3C-CH-CH-CH2-C-NH-CH2 -CH3 Common d. H3C-CH2-CH2 CH3 IUPAC H3C-CH2-CH2 -CH3 Commonarrow_forwardReagents a. CeHsCHO b. NaOH, ethanol c. Pyrrolidine, cat. H* d. H2C=CHCN e. H30* f. LDA g. ELOC(=0)CO,Et h. BRCH2CH=CH2 Na* OEt, ethanol j. Br2, H* k. K* t-BuO 1. CH2(CO2Et)2 m. heat Select reagents from the table to synthesize this compound from cyclopentanone. Enter the letters of the chosen reagents, in the order that you wish to use them, without spaces or punctuation (i.e. geda). Submit Answer Try Another Version 1 Item attempt remainingarrow_forward4. Designate R or S of the followings. Br C3H7 COOH C-H b) HS H CC2H5 с) Hас— он a) H a CHO a CH3 H. g) HO ČH3 e) OH f) b. H- -CI Br- -H :) ČH3 H- -CI OHarrow_forward
- 17. Name the following alkynes. b. a H HーC三c- H-c=c- H H H. H d. H-cEc-CH,-CH3 -c=c- H- с—н H H .CH3 f. H2Ç .CH2 c-c=CH H3C. H H-C-H CH H-C C-C- -c- C-H CH3 H2C. H H Н-с-н CH2 H CH3 I-O- O-arrow_forwardi. H H,C-C -o -CH,CH, j. H,C он 0 -Harrow_forwardChoose the correct product for each reaction. A. B. C. D. PPh3 H PPh3 || I ။arrow_forward
- 5. What is the product? A. HCI H₂C CH₂ HOCH D. A and B A. A B. B C. C D. D E. E -CH3 2NaNH₂ H₂C B. C=CH H CH3 E. A, B and C C. H₂C-CEC-CH3arrow_forwardDirections: Identify the given chemical structures as identical isomers or different compounds. Identical/ Isomers/ Chemical structure Chemical structure Different compounds CH, CH, a. H,C- -CH, CH CH b. CH H,C CH, H,C С. H,C -CH, OH H,C-CH,-CH, OH d. CH2 H,C-CH=CH2 CH2 е. H,C. H. CH CH, CH, f. H3C- CH H,C- -CH-CH, ČH, g. H,C-CH,-NH, CH3 h. H,C- -CH2 -CH2-CH, i. -CH,-CH, H,C-CH,-C 0-H H- j. H- -O -CH,-CH,-OH но CH2 -OH k. H,C-č-CHCH, H3C-CH2-C-CH3arrow_forwardComplete the following reactions My HC-O H&C KCN C. CHO Na HC-CH,-B d. H.C KMnO H.C HO CH KMrO, 0- + H,C~ H.C HI CHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
GCSE Chemistry - Differences Between Compounds, Molecules & Mixtures #3; Author: Cognito;https://www.youtube.com/watch?v=jBDr0mHyc5M;License: Standard YouTube License, CC-BY