
Organic Chemistry: Structure and Function
8th Edition
ISBN: 9781319079451
Author: K. Peter C. Vollhardt, Neil E. Schore
Publisher: W. H. Freeman
expand_more
expand_more
format_list_bulleted
Question
Chapter 8.8, Problem 8.19E
Interpretation Introduction
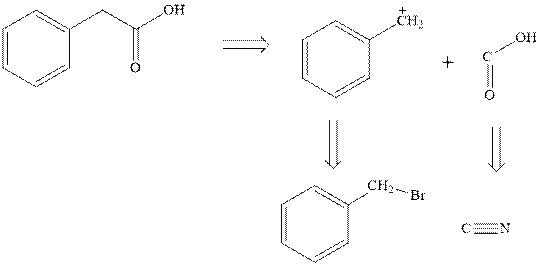
Interpretation:The retrosynthetic analysis should be applied to
Concept introduction:Retrosynthetic analysis involves transformation of target molecule into simple precursor that can be used as substrate for the synthesis of target molecule. The bonds broken generate molecules called synthons or synthetic equivalent. The latter term is designated to indicate their use as substrate in synthesis. For instance, the synthons generated in phenylacetic acid are given as follows:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
3) Beginning from acetylene and any alkyl halide needed, how would you prepare the
following compound, there may be more than one step.
HC=CH
CH;CH,CH,CH,CH,CH,CH;
An important step in one synthesis of carboxylic acids is the deprotonation of diethyl malonate and its alkyl-substituted
derivative:
Base
CH;CH2O
OCH,CH3
CH;CH,0
OCH2CH3
H2
Diethyl malonate
Base
CH;CH,0
°C
`OCH,CH3
CH;CH,O
OCH,CH3
R
Alkyl substituted diethyl malonate
NaOH can deprotonate diethyl malonate effectively, but NaOC(CH3)3 is typically used to deprotonate the alkyl-substituted
derivative. Explain why.
Identify the best reagents to complete the following reaction. Options are included.
Chapter 8 Solutions
Organic Chemistry: Structure and Function
Ch. 8.1 - Prob. 8.1ECh. 8.1 - Prob. 8.2ECh. 8.3 - Prob. 8.4TIYCh. 8.3 - Prob. 8.5ECh. 8.3 - Prob. 8.6ECh. 8.4 - Prob. 8.7ECh. 8.5 - Prob. 8.8ECh. 8.5 - Prob. 8.9ECh. 8.5 - Prob. 8.10ECh. 8.5 - Prob. 8.11E
Ch. 8.5 - Prob. 8.12ECh. 8.6 - Prob. 8.14TIYCh. 8.7 - Prob. 8.15ECh. 8.7 - Prob. 8.16ECh. 8.8 - Prob. 8.18TIYCh. 8.8 - Prob. 8.19ECh. 8.8 - Prob. 8.21TIYCh. 8 - Prob. 24PCh. 8 - Prob. 25PCh. 8 - Prob. 26PCh. 8 - Prob. 27PCh. 8 - Prob. 28PCh. 8 - Prob. 29PCh. 8 - Prob. 30PCh. 8 - Prob. 31PCh. 8 - Prob. 32PCh. 8 - Prob. 33PCh. 8 - Prob. 34PCh. 8 - Prob. 35PCh. 8 - Prob. 36PCh. 8 - Prob. 37PCh. 8 - Prob. 38PCh. 8 - Prob. 39PCh. 8 - Prob. 40PCh. 8 - Prob. 41PCh. 8 - Prob. 42PCh. 8 - Prob. 43PCh. 8 - Prob. 44PCh. 8 - Prob. 45PCh. 8 - Prob. 46PCh. 8 - Prob. 47PCh. 8 - Prob. 48PCh. 8 - Prob. 49PCh. 8 - Prob. 50PCh. 8 - Prob. 51PCh. 8 - Prob. 52PCh. 8 - Prob. 53PCh. 8 - Prob. 54PCh. 8 - Prob. 55PCh. 8 - Prob. 56PCh. 8 - Prob. 57PCh. 8 - Prob. 58PCh. 8 - Prob. 59PCh. 8 - Prob. 60PCh. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - Prob. 64PCh. 8 - Prob. 65PCh. 8 - Prob. 66P
Knowledge Booster
Similar questions
- How can you synthesize 3-methyl-1-benzenesulfonic acid and 4-methyl-1-benzenesulfonic acid starting from benzene.arrow_forwardWhat reaction conditions are needed to convert (R)-2-ethyl-2- methyloxirane to (R)-2-methylbutane-1,2-diol ?arrow_forward5. Describe an efficient synthesis of 1-methyl-1-cyclohexanol from cyclohexanol. Show the structure of the reagents, reactants and products from each step. Cyclohexanol -------> -----> 1-methyl-1-cyclohexanolarrow_forward
- CH₂-CH₂-CH₂-C-OH butanoic acid CH₂ HO–C–CH,CHCH,—CHO 3-methylpentanoic acid Name each carboxylic acid. (a) HO CH₂ CH₂ 99 - 0 B) CHICHICIO CH, Draw a condensed structural diagram for each carboxylic acid (a) Hexanoic acid (b) 3-propyloctanoic acid Iarrow_forwardAcetals are formed from the reaction of two alcohols with a carbonyl under acidic conditions. Acetal formation is faster with 1,2-ethanediol than with two methanol molecules. Choose the factor that explains the difference in reaction rates. A) The reaction with 1,2-ethanediol has a lower AH (enthalpy) of reaction. B) The reaction with 1,2-ethanediol has a higher AH (enthalpy) of reaction. C) The reaction with 1,2-ethanediol has a more favorable entropy of reaction.arrow_forward16) The best classification for the following compound is: O 0 A) aldehyde B) ester C) ketone D) carboxylic acid 17) The name of the following compound: A) phenyl ethanoate B) phenyl propanoate C) propyl benzoate D) butyl benzoate 18) Complete the following reaction: CH3COOC₂H5 + NaOH- A) CH3COCH 3 B) CH3COCH₂CH3 C) CH3COONa D) CH3COOCH₂ من + HOC₂H5.arrow_forward
- Acid-catalyzed dehydration of 3-methyl-2-pentanol gives three alkenes: 3-methyl-1-pentene, 3-methyl-2-pentene, and 3-methylenepentane. Draw the structure of the carbocation intermediate leading to the formation of 3-methyl-2-pentene.arrow_forwardHow to synthesize trans-2-methylcyclohexanol from cyclohexanone?arrow_forwardDescribe how 1-ethylcyclohexanol can be prepared from cyclohexane. You can use any inorganic reagents, any solvents, and any organic reagents as long as they contain no more than two carbons.arrow_forward
- OChem help with IUPAC names involving Ph and Bn The phenyl group (Ph-R, C6H5-R) can be formed by removing a hydrogen from benzene and attaching a substituent to where the hydrogen was removed. The benzyl group (abbv. Bn), similar to the phenyl group, is formed by manipulating the benzene ring. In the case of the benzyl group, it is formed by taking the phenyl group and adding a CH2 group to where the hydrogen was removed. Its molecular fragment can be written as C6H5CH2-R, PhCH2-R, or Bn-R. Please provide the IUPAC name for the following: (Ph)2CHC(CH3)2CC(CH2)3CH(Bn)CHOarrow_forwardWrite in the correct reagents for these transformations: a) cyclohexanol -→ chlorocyclohexane b) 2-butanone 2-butanol ward 2-butanol 2-butanone toluene - → benzoic acid nitrobenzene → anilinearrow_forwardSynthesize 4-octanol using only 1-butene as your source of carbonarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning