
Concept explainers
(a)
Interpretation:The indicated synthesis for alcohol should be evaluated as either good, not so good or worthless.
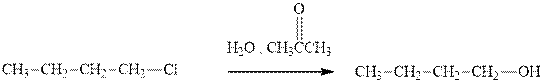
Concept introduction:Haloalkane solvolysis with ethanol, methanol or water is a typical example for unimolecuar substitution. It proceeds via two-step mechanism. The first slow step that determines rate is the removal of leaving group from the substrate haloalkane and generates a carbocation. Since the rate is only governed by substrate alone and no other nucleophile or solvent it is termed as unimolecuar substitution. The final step is attack of nucleophile on carbocation generated and formation of racemic products.
Tertiary or secondary halides undergo fastest unimolecuar substitution as they can readily form the tertiary carbocation followed by secondary and least reactive are primary.
(b)
Interpretation: The indicated synthesis for alcohol should be classified as either good, not so good or worthless.
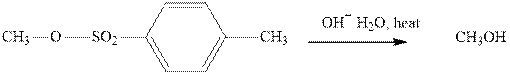
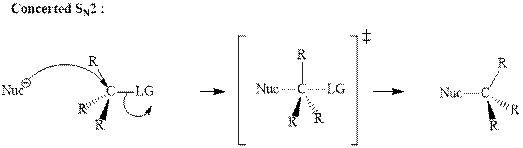
Concept introduction:Bimolecular substitution or
A general
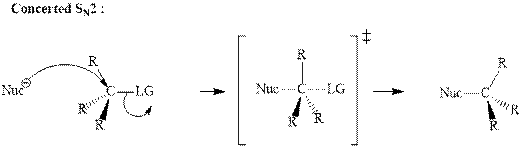
(c)
Interpretation: The indicated synthesis for alcohol should be classified as either good, not so good or worthless.
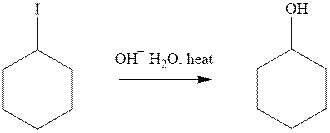
Concept introduction:Bimolecular substitution or
A general
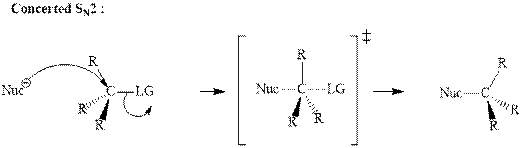
(d)
Interpretation: The indicated synthesis for alcohol should be classified as either good, not so good or worthless.
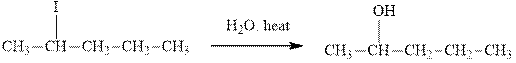
Concept introduction: Bimolecular substitution or
A general
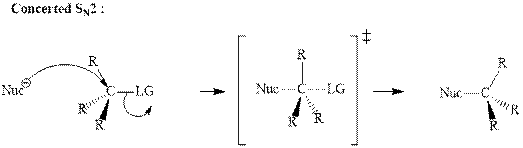
(e)
Interpretation: The indicated synthesis for alcohol should be classified as either good, not so good or worthless.
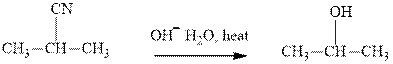
Concept introduction: Bimolecular substitution or
A general
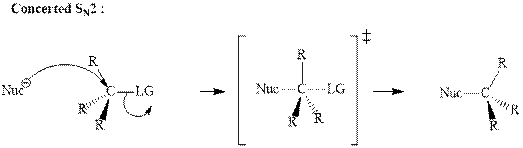
(f)
Interpretation: The indicated synthesis for alcohol should be classified as either good, not so good or worthless.
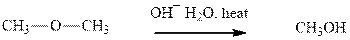
Concept introduction: Bimolecular substitution or
A general
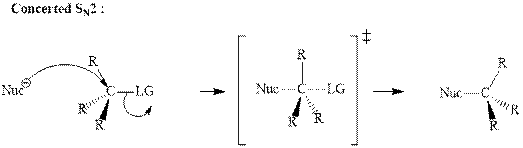
(g)
Interpretation: The indicated synthesis for alcohol should be evaluated as either good, not so good or worthless.
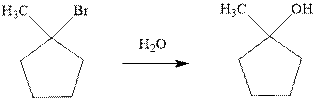
Concept introduction: Haloalkane solvolysis with ethanol, methanol or water is a typical example for unimolecuar substitution. It proceeds via two-step mechanism. The first slow step that determines rate is the removal of leaving group from the substrate haloalkane and generates a carbocation. Since the rate is only governed by substrate alone and no other nucleophile or solvent it is termed as unimolecuar substitution. The final step is attack of nucleophile on carbocation generated and formation of racemic products.
Tertiary or secondary halides undergo fastest unimolecuar substitution as they can readily form the tertiary carbocation followed by secondary and least reactive are primary.
(h)
Interpretation: The indicated synthesis for alcohol should be classified as either good, not so good or worthless.
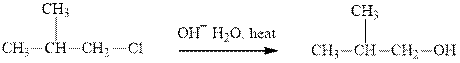
Concept introduction: Bimolecular substitution or
A general
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Organic Chemistry: Structure and Function
- An important step in one synthesis of carboxylic acids is the deprotonation of diethyl malonate and its alkyl-substituted derivative: Base CH;CH2O OCH,CH3 CH;CH,0 OCH2CH3 H2 Diethyl malonate Base CH;CH,0 °C `OCH,CH3 CH;CH,O OCH,CH3 R Alkyl substituted diethyl malonate NaOH can deprotonate diethyl malonate effectively, but NaOC(CH3)3 is typically used to deprotonate the alkyl-substituted derivative. Explain why.arrow_forwardIdentify the product, if any, that would form in each of the following reactions. 73) CH3 - CH2-CH2-OH [0] A) || CH3- CH2 - C- CH3 74) [0] B) CH 3 - CH 2 - CH -OH || CH3 CH3 - CH2 - CH Match the structural formula with the correct functional group. 75) CНз - СH2 -ОН A) alcohol 76) CH3 – CH2¤0 ICH3 B) thiol 77) CH3- CH2 - SH C) ether Select the correct name for the following. 78) CH3 - CH2 - CH2 - SH A) propanethiol 79) CH3 - CH2 - O - CH2 - CH3 B) diethyl ether 80) C) m-ethylphenol OH CH2CH3 D) 1-ethyl-3- hydroxycyclohexene E) propane sulfidearrow_forwardMixing cyclohexanol with phosphoric acid is an exothermic process, whereas the production of cyclohexene is endothermic. Construct an energy diagram showing the course of this reaction. Label the diagram with the starting alcohol, the oxonium ion (the protonated alcohol), the carbocation, and the product.arrow_forward
- Write the reagents and reaction conditions that are necessary for each of the following transformations.arrow_forward1) The carbon-oxygen double bond present in aldehydes and ketones is very polar. What does this mean and how does it arise? 2) The carbon-oxygen double bond is readily attacked by nucleophiles like cyanide ions or ammonia. (i) What do you understand by the term nucleophile? (ii) Which part of the carbon-oxygen double bond is attractive to nucleophiles? 3) Why is there a difference between aldehydes and ketones in their response to oxidizing agents such as potassium dichromate(VI) solution acidified with dilute sulfuric acid?arrow_forward4. Complete the following reactions. Only include the major products.arrow_forward
- You may want to reference (Pages 408 - 414) Section 12.3 while completing this problem. The compound frambinone has the taste of raspberries and has been used in weight loss. Its structure is HO |||-CH₂-CH, -C-CH3 ▾ Part A Identify the functional groups in frambinone. Check all that apply. ketone aromatic aldehyde ester ether phenol Submit Request Answerarrow_forwardThe overall dehydration reaction we are performing (the dehydration of cyclohexanol) is reversible and actually slightly endothermic. How can we shift the equilibrium to the product side, assuming we can not limitlessly increase the temperature? Increased magnetic stirring le Chatelier's principle Increased amount of catalyst Extended reaction timearrow_forwardDraw the structural formula of an alkene that undergoes acid-catalyzed hydration to give each of the following alcohols as the major product. More than one alkene may give each compound as the major productarrow_forward
- Identify the most important aldehyde and ketone from Section 14.4 on the basis of amount used, and list at least one characteristic for each that contributes to its usefulness.arrow_forward8) Show how the following conversions might be accomplished, including all reagents and intermediates. Br Y. OHarrow_forwardCan you prepare diisopropyl ether as the major product by heating 2-propanol in the presence of sulfuric acid? O Yes, because these conditions promote the substitution reactions. O Yes, because these conditions are highly favorable for O No, because secondary and tertiary alcohols undergo elimination reactions in these conditions. O No, because secondary and tertiary alcohols do not react in the presence of sulfuric acid.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


