
Concept explainers
Interpretation:The missing reagent required in the synthetic transformation scheme indicated should be filled sequentially.
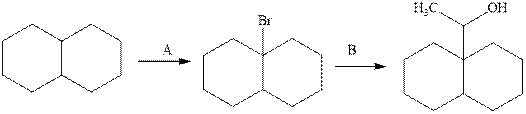
Concept introduction:Grignard reagents are
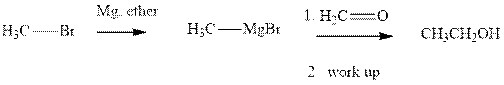
Methyl bromide reacts with magnesium ether to give Grignard reagent. This Grignard methyl magnesium bromide on treatment with formaldehyde gives corresponding alcohol. The mechanistic pathway for the latter reaction is as follows:
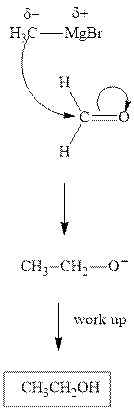
The polar carbonyl bond breaks and the polar Grignard reagent attack at electron-deficient carbon. Finally with the hydrolysis and work up the alcohol is formed.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Organic Chemistry: Structure and Function
- Several reagents and several organic structures are shown below. Construct a multistep synthetic route from the reactant 2-methyl-1-butene to the product 3-bromo-2-methyl-2-butanol by dragging the appropriate pieces into the bins. Note that each bin will hold only one item, and not every given reagent or structure will be used.arrow_forwardFill in the missing reagents, conditions, intermediates or products in each box below.arrow_forwardWhat are the necessary reagents A-D (include any necessary work-up steps) needed to produce the intermediates and final product shown in the above synthetic scheme?arrow_forward
- For each of the reactions below fill in the reagent necessary to carry out the observed transformationarrow_forwardDraw the structures of the missing reactants, intermediates, or products, in the following mechanism. Use wedges and dashes to indicate stereochemistry when appropriate.arrow_forwardFill in the missing reagent/product(s) for the following reactions.arrow_forward
- Provide reagents for the first transformation of the synthetic sequence shown below. Draw the final product of the sequence in the box.arrow_forwardDecide which of the following molecules these statements belong to. Each molecule is only used as answer once. Br A B Br C Br D This molecule has the highest reaction rate of [Choose ] all in an SN2 reaction. [Choose ] This molecule has the highest reaction rate in an D SN1 reaction. A This molecule can neither react by an SN1 nor an SN2 mechanism. E B This molecule can react by an SN1, E1 or E2 mechanism but will never react by an SN2 mechanism. C F This moelcule can react my an SN2 or E2 mechanism but will not react by an SN1 or E1 mechanism. [Choose] This molecule reacts rapidly by either an SN1 or [Choose ] an SN2 mechanism. له Br Br E F Brarrow_forwardAbove each arrow, supply the reagent(s)/reaction conditions that is(are) needed for each of the following (Only one reaction is necessary for each transformation.) transformations. Br SHarrow_forward
- The following transformation can be accomplished by reactions we have studied in this chapter and Chapter 20. Name the type of reaction used in each step.arrow_forwardThe following molecules are subject to substitution (SN1 or SN2) reaction conditions. a) Identify if the leaving group (-Br) is attached to a 3°, 2°, 1°, or methyl Carbon. b) Rank the molecule with respect to their SN1 reactivity, with 1 being the fastest and 4 being the slowest. c) Rank the molecule with respect to their SN2 reactivity, with 1 being the fastest and 4 being the slowest. Br Br CH3B Br a. type of Carbon on C-Br b. SN1 reactivity (1 fastest, 4 slowest) c. SN2 reactivity (1 fastest, 4 slowest)arrow_forwardFor each reaction shown below draw the product. make sure to include any stereochemistry.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

