
Concept explainers
(a)
Interpretation:Synthesis of
Concept introduction: The carbonyl bond is polar with the partial positive charge on carbon and partial negative charge on oxygen as illustrated below.
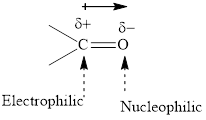
Thus it can undergo hydride addition at carbon and proton addition at oxygen. Certain reagents that are useful for such hydride addition at carbonyl carbon include sodium borohydride, lithium aluminum hydride. The boron and lithium in these reagents tend to push the electron of
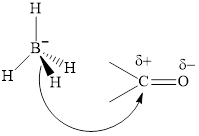
(b)
Interpretation:Synthesis of
Concept introduction:The alcohols are best synthesized from on the basis of retrosynthetic analysis with the aid of Grignard reagents.
The carbonyl bond is polar with partial positive charge on carbon and partial negative charge on oxygen as illustrated below.
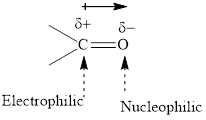
Grignard reagents are
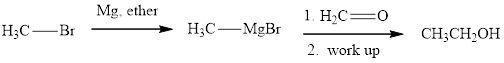
Methyl bromide reacts with magnesium ether to give Grignard reagent. This Grignard methylmagnsium bromide on treatment with formaldehyde gives corresponding alcohol. The mechanistic pathway for the latter reaction is as follows:
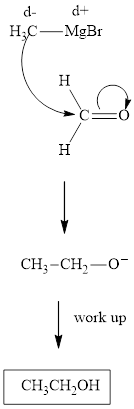
The polar carbonyl bond breaks and the polar Grignard reagent attack at electron-deficient carbon. Finally with the hydrolysis and work up the alcohol is formed.
(c)
Interpretation: Synthesis of
Concept introduction: The alcohols are best synthesized from on the basis of retrosynthetic analysis with the aid of Grignard reagents.
The carbonyl bond is polar with the partial positive charge on carbon and partial negative charge on oxygen as illustrated below.
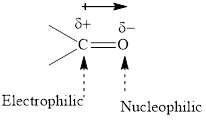
Grignard reagents are alkyl magnesium halides obtained from the treatment of haloalkane with magnesium in the presence of dried ether conditions. Theses reagent are a useful precursor for the quick synthesis of a variety of organic compounds For example, the reaction of Grignard reagent with aldehyde or ketone generates alcohols as illustrated below.
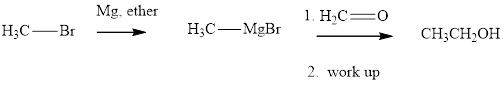
Methyl bromide reacts with magnesium ether to give Grignard reagent. This Grignard methylmagnsium bromide on treatment with formaldehyde gives corresponding alcohol. The mechanistic pathway for the latter reaction is as follows:
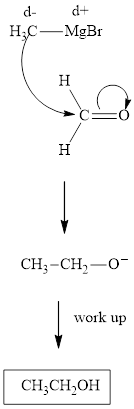
The polar carbonyl bond breaks and the polar Grignard reagent attack at electron-deficient carbon. Finally with the hydrolysis and work up the alcohol is formed.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Organic Chemistry: Structure and Function
- Draw the structure of each of the following molecules. (a) pentanoic anhydride; (b) hexanoic propanoic anhydride; (c) ethanoic 3-methylpentanoic anhydridearrow_forwardDraw the organic product formed when the followingcompounds undergo a substitution reaction: (a) acetic acid and1-hexanol; (b) propanoic acid and dimethylamine; (c) ethanoicacid and diethylamine.arrow_forwardFor each of the following trivial names, draw the structure and write the correct IUPAC name. (a) trichloroacetic anhydride; (b) valeric anhydride; (c) acetic butyric anhydridearrow_forward
- Synthesize each compound from benzonitrile (C6H5CN) as the only organic starting material; that is, every carbon in the product must originate in benzonitrile.arrow_forwardDraw the alcohol you’d oxidize to produce (a) 2-methyl-propanal; (b) 2-pentanone; (c) 3-methylbutanoic acid.arrow_forward(a) Draw a specific organic reaction for the hydrohalogenation addition reaction of alkenes. (b) Also write the reagents involved in the reaction (c) and name and identify its major and minor product by writing its IUPAC name.arrow_forward
- 1) The acid-catalyzed dehydration of 2-methyl-2-butanol yields two alkene products, what are the names of the two alkenes? 2) which of the two alkenes is the major product?arrow_forward(a) Draw a specific organic reaction for the hydration addition reaction of alkenes. (b) Also write the reagents involved in the reaction (c) and name and identify its major and minor product by writing its IUPAC name.arrow_forwardDraw the molecule that corresponds to each IUPAC name. (a) 3,3-dipropoxypentan-1-amine; (b) 2,3,4-trichlorocyclohexanol; (c) 3-cyclopropylpentan-1-ol; (d) 3-(1-methylethyl)cycloheptanaminearrow_forward
- 5. Synthesis: Synthesize the following compound from cyclohexane, any carbon pieces that are three carbons or less (can include other functional groups), and any inorganic reagents.arrow_forwardA) An Alcohol X has the structure of (CH,), C(OH)CH(CH,),. i) State the IUPAC name of alcohol X. i) Outline the mechanism for the reaction occurring when alcohol X is converted into 2,3-dimethylbut-2-ene in the presence of a strong acid. i) Name the reaction in part ii). iv) Draw the structure and state the IUPAC name of an isomer of 2,3-dimethyl-2- butene which is also formed in the reaction. v) Explain why two products are obtained. vi) Write an equation for the reaction between alcohol X and ethanoyl chloride. vii) Outline a mechanism for this reaction, using ROH to represent the alcohol in the mechanism.arrow_forwardComplete the following syntheses – they may be two- or three-step processes. Include any necessary catalysts or reaction conditions. b) Prepare ethanoic acid from ethenearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





