
Organic Chemistry: Structure and Function
8th Edition
ISBN: 9781319079451
Author: K. Peter C. Vollhardt, Neil E. Schore
Publisher: W. H. Freeman
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 1, Problem 44P
Interpretation Introduction
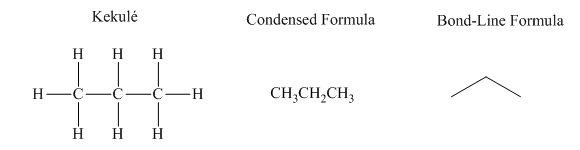
Interpretation:The indicated condensed formulasand Kekule structures should be drawn as bond-line formulas.
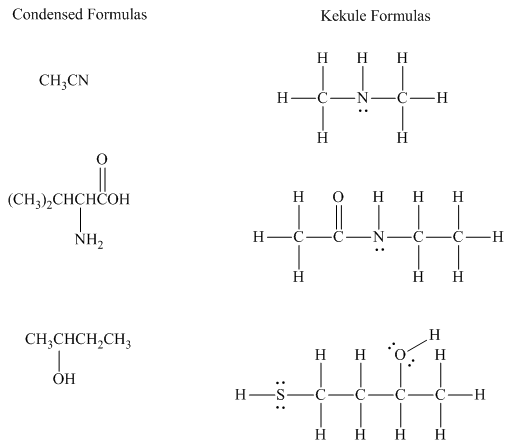
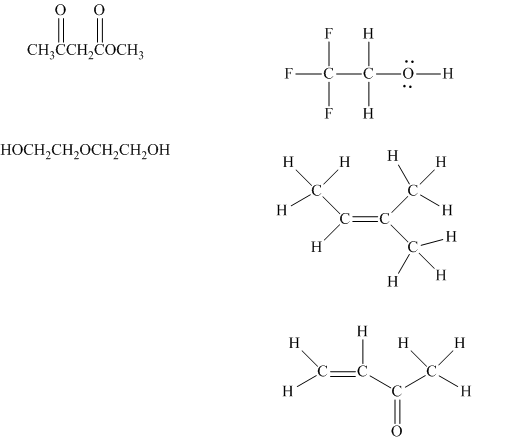
Concept introduction: There are three ways of representation of molecular structures. The hashed and wedged formulas have three types of bonds. The bond above the plane is represented by bold hash while bonds behind the plane are represented by wedge. The bonds in plane are represented by normal bonds.
Bond-line formula depicts the zig-zag framework of carbon chain with attached substituents. The terminal position represents a methyl
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Four major contributing resonance structures are possible for the given cation, which is the intermediate o complex of an
electrophilic aromatic substitution involving phenol and bromine. Two structures are given but are incomplete. Complete the
given structures by adding nonbonding electrons and formal charges. Draw the remaining structures (in any order), including
nonbonding electrons and formal charges.
Complete structure A.
Complete structure B.
: 0 H
:0-
Br :
Br
Provide an arrow-pushing mechanism to explain the formation of benzoic acid from bromobenzene. (Use line drawings with lone pairs and formal charges)
Complete the scheme, write formulas:
S
CISO3H
- 15°C
konc. HNO3
H₂O
A
B
C4H3NSO₂
40°C
to
Chapter 1 Solutions
Organic Chemistry: Structure and Function
Ch. 1.3 - Prob. 1.1ECh. 1.3 - Prob. 1.2ECh. 1.3 - Prob. 1.3ECh. 1.3 - Prob. 1.4ECh. 1.3 - Prob. 1.5ECh. 1.4 - Prob. 1.6ECh. 1.4 - Prob. 1.8TIYCh. 1.5 - Prob. 1.9ECh. 1.5 - Prob. 1.11TIYCh. 1.6 - Prob. 1.12E
Ch. 1.6 - Prob. 1.13ECh. 1.7 - Prob. 1.15TIYCh. 1.8 - Prob. 1.16ECh. 1.8 - Prob. 1.18TIYCh. 1.9 - Prob. 1.19ECh. 1.9 - Prob. 1.20ECh. 1.9 - Prob. 1.21ECh. 1.9 - Prob. 1.22ECh. 1 - Prob. 25PCh. 1 - Prob. 26PCh. 1 - Prob. 27PCh. 1 - Prob. 28PCh. 1 - Prob. 29PCh. 1 - Prob. 30PCh. 1 - Prob. 31PCh. 1 - Prob. 32PCh. 1 - Prob. 33PCh. 1 - Prob. 34PCh. 1 - Prob. 35PCh. 1 - Prob. 36PCh. 1 - Prob. 37PCh. 1 - Prob. 38PCh. 1 - Prob. 39PCh. 1 - Prob. 40PCh. 1 - Prob. 41PCh. 1 - Prob. 42PCh. 1 - Prob. 43PCh. 1 - Prob. 44PCh. 1 - Prob. 45PCh. 1 - Prob. 46PCh. 1 - Prob. 47PCh. 1 - Prob. 48PCh. 1 - Prob. 49PCh. 1 - Prob. 50PCh. 1 - Prob. 51PCh. 1 - Prob. 52PCh. 1 - Prob. 53PCh. 1 - Prob. 54PCh. 1 - Prob. 55PCh. 1 - Prob. 56PCh. 1 - Prob. 57PCh. 1 - Prob. 58P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Specify the local electron geometries about the atoms labelled a-d. Unshared electron pairs affect local geometry and are included in the structural formula.For each atom enter one of the following: tetrahedral, square planar, trigonal planar, trigonal pyramidal, or linear.arrow_forwardWhich structure is correct? Please include the arrow pushing & resonance to guide mearrow_forwardIdentify the name of the following bond-line structuresarrow_forward
- Write an equation to show the reaction between ethanol, C2H5OH and methyllithium, CH3 Draw all non-bonding electrons and show electron flow with curved arrows.arrow_forwardBriefly explain why the hydrocarbon shown below possesses a significant dipole moment. Use diagrams as needed to illustrate/clarify your answer.arrow_forwardSpecify the local electron geometries about the atoms labelled a-d. Unshared electron pairs affect local geometry and are included in the structural formula. For each atom enter one of the following: tetrahedral, square planar, trigonal planar, trigonal pyramidal, or linear. H. d (a) (b) (c) (d)arrow_forward
- An electrostatic potential map of calicene is shown below. a) Both the electrostatic potential map and its significant dipole moment indicate that calicene is an unusually polar hydrocarbon. Which of the dipolar resonance forms, structure A or structure B, better corresponds to the electron distribution in the molecule? Select the single best answer. b) Which one of the following structures should be stabilized by resonance to a greater extent? Select the single best answer.arrow_forwardHow much of heat is produced when 125 g of acetylene undergoes combustion? Information given ( std enthapy of combustion of acetylene is 1301 KJ) (Some places I have seen this being solved using bond dissociation enthalpy, however the simple moles method is what I would prefer) Thanksarrow_forwardUse the arrows to account for the bond breaking and electron pushing belowarrow_forward
- Write the condense structure of the following compound 1.Cyclobutanone 2. Propanenitrile 3. Pentanamidearrow_forwardProvide the IUPAC name for this structure: Type your answer and submit X₂ X² Ω· Write your response here.…... ОН ОНarrow_forward19.) The thiocyanate ion (SCN-) has thee resonance structures. Each follows the octet rule (#1) has a sulfur to carbon single bond and a carbon to nitrogen triple bond. (#2) has a sulfur to carbon triple bond and a carbon to nitrogen single bond. (#3) has a sulfur to carbon double bond and a carbon to nitrogen double bond. Which structure is the "best" one using formal charge arguments? Group of answer choices 2 all three are equal 1 3 1 and 2 are equalarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning