
Organic Chemistry: Structure and Function
8th Edition
ISBN: 9781319079451
Author: K. Peter C. Vollhardt, Neil E. Schore
Publisher: W. H. Freeman
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 1, Problem 56P
Interpretation Introduction
Interpretation:The orbitals involved in formation of bond showed by arrow should be chosen.
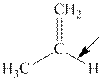
Concept introduction:In accordance with VSEPR model, if four bonds are present around the central atom the hybridization associated is
Similarly, if three bonds are present around the central atom then hybridization associated is
The table that relates the hybridization with bond angles is as follows:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw an orbital picture of propene. The bond between and is formed by the overlap of which types of orbitals?
a. sp2 and sp2
b. sp3 and sp3
c. sp2 on Ca and sp3 on Cb
d. sp3 on Ca and sp2 on Cb
The double bond between and is formed by what?
a. two sigma bonds
b. two pi bonds
c. overlap of a p orbital on Cb with a sp2 orbital on Cc.
d. overlap of two p orbitals plus overlap of two sp2 orbitals
The arrow in the structure points to a bond that is formed by
a. overlap between s and sp2 orbitals
b. overlap between two sp orbitals
c. overlap between two sp3 orbitals
d. overlap between sp and sp2 orbitals
e. overlap between sp2 and sp3 orbitals
21. What atomic orbitals of carbon are used for the formation of the sigma bond
component of the C-C triple bond?
a. 2sp atomic orbitals
b. 2sp atomic orbitals
C. 2p atomic orbitals
d. 2s atomic orbitals
e. 2sp´ atomic orbitals
Chapter 1 Solutions
Organic Chemistry: Structure and Function
Ch. 1.3 - Prob. 1.1ECh. 1.3 - Prob. 1.2ECh. 1.3 - Prob. 1.3ECh. 1.3 - Prob. 1.4ECh. 1.3 - Prob. 1.5ECh. 1.4 - Prob. 1.6ECh. 1.4 - Prob. 1.8TIYCh. 1.5 - Prob. 1.9ECh. 1.5 - Prob. 1.11TIYCh. 1.6 - Prob. 1.12E
Ch. 1.6 - Prob. 1.13ECh. 1.7 - Prob. 1.15TIYCh. 1.8 - Prob. 1.16ECh. 1.8 - Prob. 1.18TIYCh. 1.9 - Prob. 1.19ECh. 1.9 - Prob. 1.20ECh. 1.9 - Prob. 1.21ECh. 1.9 - Prob. 1.22ECh. 1 - Prob. 25PCh. 1 - Prob. 26PCh. 1 - Prob. 27PCh. 1 - Prob. 28PCh. 1 - Prob. 29PCh. 1 - Prob. 30PCh. 1 - Prob. 31PCh. 1 - Prob. 32PCh. 1 - Prob. 33PCh. 1 - Prob. 34PCh. 1 - Prob. 35PCh. 1 - Prob. 36PCh. 1 - Prob. 37PCh. 1 - Prob. 38PCh. 1 - Prob. 39PCh. 1 - Prob. 40PCh. 1 - Prob. 41PCh. 1 - Prob. 42PCh. 1 - Prob. 43PCh. 1 - Prob. 44PCh. 1 - Prob. 45PCh. 1 - Prob. 46PCh. 1 - Prob. 47PCh. 1 - Prob. 48PCh. 1 - Prob. 49PCh. 1 - Prob. 50PCh. 1 - Prob. 51PCh. 1 - Prob. 52PCh. 1 - Prob. 53PCh. 1 - Prob. 54PCh. 1 - Prob. 55PCh. 1 - Prob. 56PCh. 1 - Prob. 57PCh. 1 - Prob. 58P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Show how a dxz. atomic orbital and a pz, atomic orbital combine to form a bonding molecular orbital. Assume the x-axis is the internuclear axis. Is a or a molecular orbital formed? Explain.arrow_forwardIn terms of the molecular orbital model, which species in each of the following two pairs will roost likely be the one to gain an electron? Explain. a. CN or NO b. O22+ or N22+arrow_forwardIdentify any carbon atoms that change hybridization and the change in hybridization during the reactions in Exercise 20.51.arrow_forward
- Sketch the geometry of (a) tans-[Cu(Br)2(H2O)4] (b) [Zn(OH)2I22-](tetrahedral) (c) [Pt(NH3)42+](square planar) (d) cis-[Ni(H2O)2(C2O4]22- (e) [Au(CN)(NH3)]arrow_forwardExplain what is wrong with each of the following statements. a. “A bond is a double bond. b. “A bond consists of four electrons, one in each of the four p orbitals involved in the bond. c. “A bond is twice as strong as a bond because it consists of two orbital overlaps insteadof just one.”arrow_forwardWhich of the following correctly describes a pi bond according to valence bond theory? Select one: O A. A pi bond is formed between adjacent atoms when two p orbitals, co- linear with the bond axis, meet "head on". OB. A pi bond is formed when an s orbital overlaps with a hybrid orbital. OC. A pi bond is formed between adjacent atoms when hybrid orbitals, co- linear with the bond axis, overlap. OD. A pi bond is formed when two adjacent, parallel p orbitals overlap along their sides.arrow_forward
- Chapter 22 Organic Chemistry 1. Consider the line-angle drawing of a molecule given below. A. What is the molecular formula of the molecule? (1 point) B. For each the circled atom-in the molecule, what is the hybridization, electron geometry (EG), and molecular geometry (MG)? (3 points) HO A B NH CH3 Carrow_forwardWhich hybridized orbital of the carbon interact with the s orbital of the hydrogen to form the highlighted bond? A. 2sp atomic orbitals (Orbital atom 2sp) B. 2sp2 atomic orbitals (Orbital atom 2sp2) C. 2p atomic orbitals (Orbital atom 2p) D. 2sp3 atomic orbitals (Orbital atom 2sp3)arrow_forwardH-Ņ-C=C-H H. a. The bond between nitrogen and hydrogen is due to overlap of a(n) orbital. orbital and a(n) b. The lone pair on nitrogen is in what type of atomic orbital? c. The bond between N and C is due to overlap of a(n) orbital and a(n) orbital. d. The T-bonds between the two carbon atoms are due to the overlap of orbitals.arrow_forward
- 4. Draw the molecular orbital diagram for acetic acid. Label the hybridized orbitals sp, sp2, sp3, s, p. Draw in electrons pairs when needed. Indicate sigma and pi bonds. Hi sps spe Sp 3 o lbonds =6+1=7 %3D TY bnds = 1arrow_forwardPlatinum hexafluoride is an extremely strong oxidizing agent. It can even oxidize oxygen, its reaction with O2 giving O2+PtF6−.a. Sketch the molecular orbital energy level diagram for the O2+ ion.b. How many net σ and π bonds does the ion have?c. What is the O-O bond order?d. How has the bond order changed on taking away electrons from O2 to obtain O2+?e. Is the O2+ ion paramagnetic?arrow_forward10 2. Draw the four n-molecular orbitals for the molecule butadiene, drawn below. Label the four orbitals as bonding, non-bonding or antibonding. Which of the four orbitals are occupied? Which is the LUMO and which is the HOMO? sib laidio mlunal 4 Pi electrons by 88- D gilnoo slengoggu or o ed bos 2olunolom od om baod 1915b owi ni 29luoslom pa sbuloni ot olduob orb juods ohib owi ow nisiqxo HSG CH3 88 88 OCH-CH? CCICHCCHCH?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY