
(a)
Interpretation: Out of the substrates for the below reactions the one pair that will give a higher
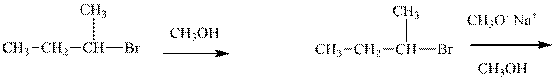
Concept introduction: Carbocation formation is relatively slower than acid-base reactions. Carbocations generated from
Analogous to the case that strong nucleophiles are more favored to react via
(b)
Interpretation: Out of the substrates for the reactions the one pair that will give a higher
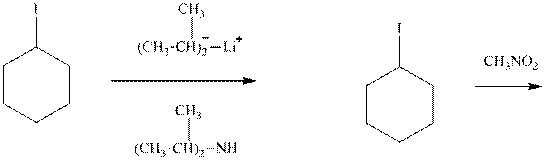
Concept introduction: Carbocation formation is relatively slower than acid-base reactions. Carbocations generated from alkyl halides have two fates; they can be either trapped by nucleophiles to give substitution product or may deprotonated to yield small amount of alkene. Stronger the base more is the probability of elimination over substitution over elimination. Further, if still higher concentration is employed reaction proceeds via bimolecular elimination. On the other hand, weak base waits until the carbocation is formed and the type of elimination with relatively weak base is two-step elimination or
Analogous to the case that strong nucleophiles are more favored to react via
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Organic Chemistry: Structure and Function
- When hydroxide is used as the base to carry out an E2 reaction on a vinylic halide, the reaction usually needs to be heated significantly. As shown below, such a reaction involving the E isomer typically requires much higher temperatures. Why is this so? Z isomer E isomer Br КОН КОН 70 °C 200-230 °C Br Br 70% 67%arrow_forward1. Compare and contrast SN1, SN2, E1 and E2 reactions. What are some obvious similarities and differences between each reaction pathway? What are the requirements for each reaction to work? Discuss why SN2 is in direct competition with E2 while SN1 is in direct competition with the El reaction pathway. What is the rate expression for each? Which Which reactions are step-wise? reactions are concerted? rode Which reactions are stereospecific and what is the stereospecificity of each that are? How do the variables below help influence which reaction pathway will lead to the major product formed? a. Substrate (substitution pattern and leaving group ability) b. Nucleophile/base strength (weak or strong) and size (unhindered or hindered) c. Solvent (polar-aprotic or polar-protic) H d. Temperature (low/ambient temps vs. higher temps) Hass of bel rai CHT Sodio Jan 201 impotavom musela or wolle simple ofer 100 HOOGM 08.1 Oroor SM o viso ( 1arrow_forward11) For each of the following reactions: Indicate whether elimination will occur via an E2 or El reaction. Explain your reasoning, and give the major elimination product of each reaction, indicating the major geometrical isomerism: i) H₂C CH3 CH₂O DMSO ii) H3C- Br Br CH3 H₂Oarrow_forward
- What product is expected from each of the following reactions. Write step by step mechanism for reaction.arrow_forwardPredict the major and minor products that would obtained from each of the following reactions (Sn1, Sn2, E1,E2) by which product is formedarrow_forward4) One of the diastereomeric cyclohexyl bromide starting materials below will readily undergo an E2 reaction while the other one will not. Fill in the requested information in boxes and lines indicated including the structure of the alkene product that does undergo an E2 reaction (use scratch paper first please). draw the expected major alkene product draw lowest E chair conformations Me. Ме Me EtO: Na Me "Ме :Br: Ме. Ме Me Me" Ме EtO: Na :Br: Required arrangement of H and LG (Br) for E2? (no acronyms). What is the rate law for and E2? On the template at left, draw all o & o* orbitals involved in the allowed E2 process.arrow_forward
- What product or products are produced in each of the following reactions? By what mechanism ( SN1, SN2,E1 or E2 ) does each react? Show the resulting products as single product, main product and by-product.arrow_forwardTell whether the given reaction will occur via SN1, SN2, E1, or E2arrow_forwardIn each reaction box, place the best reagent and conditionss from the list below CH2CN 1) 2) 3) Zn(Hg), HC NH3 NaNH2 Mg, Et2O NaBH4, CH3CH2ОН CH3NH2 Br2, FeBr3 CH3CN PBr3, pyridine H2SO4 NaCN CH3MgBr Н2, Ptarrow_forward
- For each of the questions below – determine whether the following combinations of reactants, will react by substitution (SN1 or SN2 mechanism), elimination (E1 or E2 mechanism) or will not react under the conditions given. draw the final structure and explain the logic for each decision. There may be multiple possible options for some reactions. You must include all 4 aspects of these reactions, substrate, nucleophile, solvent and leaving group in the decisionarrow_forwardComplete the table below indicating what is important in each category for an E1 or E2 reaction. E1 Rate Law Best Solvent Alkene geometry outcome How does electrophile substitution affection the reaction? Does leaving group ability affect the rate? Does base strength affect the rate? Does/how does base type affect product structure? E2arrow_forwardWhich of the reactants in the picture will undergo the fastest SN1 reaction in ethanol? O Darrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

