
Interpretation:The structure of most likely product for the indicated reaction should be written.

Concept introduction: Carbocation formation is relatively slower than acid-base reactions. Carbocations generated from
Unimolecuar substitution or
A general
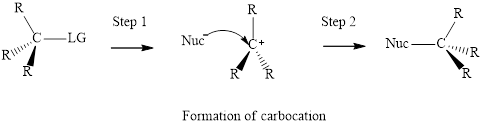
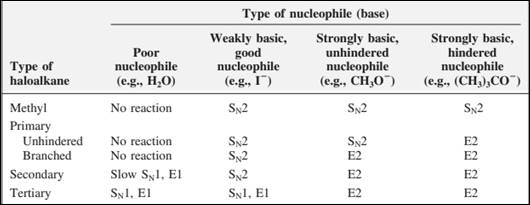
The most likely mechanisms for different kinds of alkyl halides reactions with various nucleophiles is given as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Organic Chemistry: Structure and Function
- Bicyclo-2,5-heptadiene can be prepared in two steps from cyclopentadiene and vinyl chloride. Provide a mechanism for each step.arrow_forwardThe following sequence of steps converts (R)-2-octanol to (S)-2-octanol. Propose structural formulas for intermediates A and B, specify the configuration of each, and account for the inversion of configuration in this sequence.arrow_forwardAlcohols are important for organic synthesis, especially in situations involving alkenes. The alcohol might be the desired product, or the OH group might be transformed into another functional group via halogenation, oxidation, or perhaps conversion to a sulfonic ester derivative. Formation of an alcohol from an alkene is particularly powerful because conditions can be chosen to produce either the Markovnikov or non-Markovnikov product from an unsymmetrical alkene. Using your reaction roadmap as a guide, show how to convert 4-methyl-1-pentene into 5-methylhexanenitrile. You must use 4-methyl-1-pentene and sodium cyanide as the source of all carbon atoms in the target molecule. Show all reagents needed and all molecules synthesized along the way.arrow_forward
- Alkynes do not react directly with aqueous acid as do alkenes, but will do so in the presence of mercury(II) sulfate as a Lewis acid catalyst. The reaction occurs with Markovnikov regiochemistry, so the OH group adds to the more highly substituted carbon and the H adds to the less highly substituted carbon. The initial product of the reaction is a vinyl alcohol, also called an enol. The enol immediately rearranges to a more stable ketone via tautomerization. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions -X티 Hö: H-O -CH3 -CH3 H30*arrow_forwardElimination of HBr from 2-bromobutane affords a mixture of 1-butene and 2-butene. With sodium ethoxide as base, 2-butene constitutes 81% of the alkene products, but with potassium tert-butoxide, 2-butene constitutes only 67% of the alkene products. Offer an explanation for this difference.arrow_forwardWhen 2-iodo-1,4-dimethylcyclohexane is heated in acetic acid, CH3COOH, a mixture of substitution and elimination products is obtained. Provide structures for all possible products, writing [not drawing] the name of the mechanism by which each one is formed.arrow_forward
- Write a mechanism that accounts for the formation of ethyl isopropyl ether as one of the products in the following reaction. CI OEt HCI EtOH Write the mechanism for step one of this reaction. Show lone pairs and formal charges. Only the acidic hydrogen should be drawn out with a covalent bond. Write the mechanism for step two of this reaction (where the product of step one reacts with the solvent, ethanol). Show lone pairs and formal charges. Only the acidic hydrogen should be drawn out with a covalent bond. Write the mechanism for the last step of this reaction (formation of ethyl isopropyl ether). Show lone pairs and formal charges. Only the acidic hydrogen should be drawn out with a covalent bond. CI will act as the base in this reaction.arrow_forwardTreatment of propadiene (an allene) with hydrogen bromide produces 2-bromopropene as the major product. This suggests that the more stable carbocation intermediate is produced by the addition of a proton to Br HBr. H2C=C=CH, H3C CH2 a terminal carbon rather than to the central carbon. Propadiene 2-Bromopropene (a) Draw both carbocation intermediates that can be produced by the addition of a proton to the allene. (b) Explain the relative stabilities of those intermediates. Hint: Draw the orbital picture of the intermediates and consider whether the CH, groups in propadiene are in the same plane.arrow_forwardIn an attempt to prepare propylbenzene, a chemist alkylated benzene with 1-chloropropane and aluminum chloride. However, two isomeric hydrocarbons were obtained in the proportion of 2: 1. What is the main product? How did this come about? Explain showing the mechanism and nomenclature of the reaction compoundsarrow_forward
- The optically active (2R)-2-phenyl-2-butanol reacts in hydrochloric acid to form haloalkanes and alkenes. The substitution reaction is reported to occur with 100% racemization. Give the structures of the enantiomers that form during the substitution and indicate how much of each is formed. Propose a mechanism for the substitution reaction to yield the R product. What reagents would you use to achieve 100% retention of configuration. Two geometric isomers are obtained during the elimination reaction. Explain mechanistically which alkene will be the main product.arrow_forwardGive the products, if any, of the benzene + 2 CH3Cl + AlCl3 reaction:arrow_forwardThe reaction of methylpropene with HBr, under radical conditions, gives two intermediates. Propose a mechanism for the formation of the two products. Propose a mechanism for the following reaction and use electronic factors to account for the formation of a major product: CH2 CH2Br N-Bromosuccinimide (NBS) ho, CCI4 Draw the structure of an antioxidant, Vitamin E free radical and use resonance structures o account for its stability.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

