
Concept explainers
Interpretation:A reasonable rate expression corresponding to chlorination of methane reaction should be proposed.
Concept introduction:The mechanism for monochlorination comprises of three stages illustrated as follows:
Step 1- Initiation via homolytic cleavage of
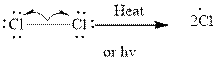
Step 2: Propagation: In the first of the propagation steps
radical generated from step 1 abstracts hydrogen radical from methane as follows:
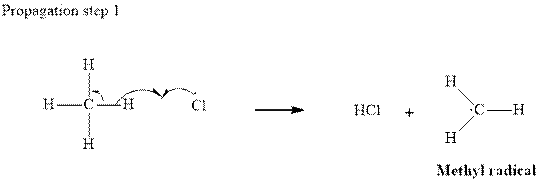
In subsequent propagation step chloromethyl radical abstracts
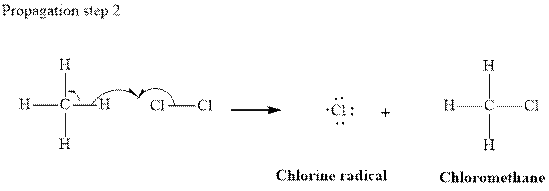
Step3: Termination: Radicals generated in propagation steps get quenched upon the combination with one another. Thus termination steps are essentially the radical− radical combination illustrated as follows:
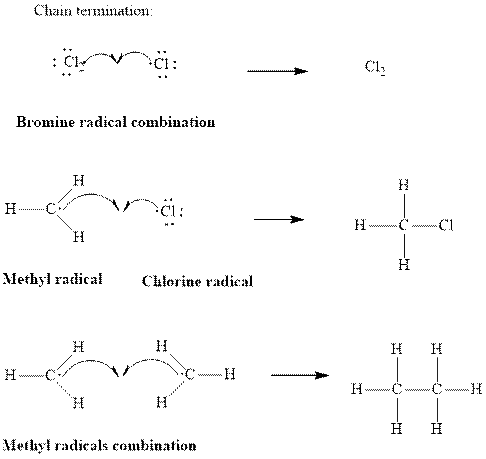
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Organic Chemistry: Structure and Function
- Page 2 of 3 D Predict the following reactions: 1. DIBAH, toluene, CH3(CH2)10COCH, 2. H₂O CH3 CrO3 H₂O* CH3CH2CHCH2CHCH2CH CH3 OH CH3CHCH2CH2CH H,C CH + C 1. (CH),CHCH,MgBr, BO 2.H,O 3 > A tv♫♫ S MacBook Airarrow_forwardWhich product is likely NOT to form in the reaction in Figure 10-9? OA B ОС ODarrow_forward5. Using curved arrows show the mechanism for the hydrohalogenation for the following reaction. Show how the two possible intermediate products are formed and explain why only one product proceeds to form the product. Clearly indicate attack of electrons and full and partial (8) charges. H-Br Br H-Br Product Intermediate Productsarrow_forward
- NBS heat Draw the molecule(s) on the canvas by choosing buttons from the Tools (for bonds and charges), A toolbars. H± 12D EXP. CONT. H C N O S F CI Br Br P F × Incorrect; Try Again; 2 attempts remaining Note that, in the second propagation step, bromine is attacked by the least sterically hindered carbon ra Δ A. Submit Previous Answers Request Answerarrow_forwardPart 3: Reaction Review Provide the missing REACTANT, REAGENT, er ORGANIC Product for cach reaction HBr но н Na Cr0, H;SO, H-0 но OHarrow_forwardIs it possible to achieve this reaction directly? Explain your reasonsarrow_forward
- Given the product/s of reaction, provide the reactants a. Н-С—С—Н b. -CH;Br + HBr С. CH,CH, Brarrow_forwardQUESTION 6 For problems 6-14, complete the reactions by filling the boxes with the necessary chemical reagent(s)/reaction conditions or major products. Provide reagent(s)/reaction conditions that would produce the major product most efficiently. Assume aqueous workups are performed if needed. ОН HO HO HO Но OH a. NaOH b. HBr O c. NABH4 Od. Ag20, H2о, NH4ОН O e. CrO3, H2O, H2SO4 O f. H2SO4 O g. LIAIH4 O h. H20 0i Crоз, НC, руridinearrow_forwardS + Ozle) → SO26) 2 SO2e) + 02 ) → 2 SO31@) SO3e) + H2SO4laq) → H;S;O7(aq) H;S;O7(a) + H2O) → 2 H;SO(aq) (a) What is the oxidation number of S on the products of the first and second equations above? (b) What is being reduced in the second equation? (c) H2S,0, is known as oleum. What is the oxidation state of sulphur in this compound? (d) Is the fourth equation a redox equation? Explain your answer.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
