
Concept explainers
(a)
Interpretation:
The step by step mechanismfor E1 process for the following reaction should be written.
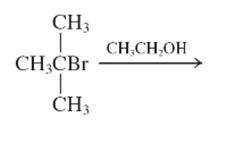
Concept Introduction:
A type of organic reaction in which two substituents get eliminated from a given molecule by one step mechanism or two step mechanism is known as elimination reaction. The reaction which occurs in one step is known as E2 reaction whereas the reaction which occurs in two steps is known as E1 reaction.
In E1 reaction, elimination of HX substituent takes place and produces a double bond. It is a unimolecular reaction.
(b)
Interpretation:
The step by step mechanismfor E1 process for the following reaction should be written.
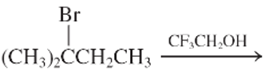
Concept Introduction:
A type of organic reaction in which two substituents get eliminated from a given molecule by one step mechanism or two step mechanism is known as elimination reaction. The reaction which occurs in one step is known as E2 reaction whereas the reaction which occurs in two steps is known as E1 reaction.
In E1 reaction, elimination of HX substituent takes place and produces a double bond. It is a unimolecular reaction.
(c)
Interpretation:
The step by step mechanismfor E1 process for the following reaction should be written.
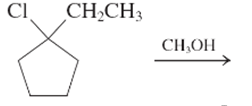
Concept Introduction:
A type of organic reaction in which two substituents get eliminated from a given molecule by one step mechanism or two step mechanism is known as elimination reaction. The reaction which occurs in one step is known as E2 reaction whereas the reaction which occurs in two steps is known as E1 reaction.
In E1 reaction, elimination of HX substituent takes place and produces a double bond. It is a unimolecular reaction.
(d)
Interpretation:
The step by step mechanismfor E1 process for the following reaction should be written.
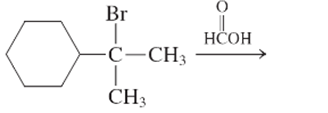
Concept Introduction:
A type of organic reaction in which two substituents get eliminated from a given molecule by one step mechanism or two step mechanism is known as elimination reaction. The reaction which occurs in one step is known as E2 reaction whereas the reaction which occurs in two steps is known as E1 reaction.
In E1 reaction, elimination of HX substituent takes place and produces a double bond. It is a unimolecular reaction.
(e)
Interpretation:
The E1 product for the following reaction should be written.
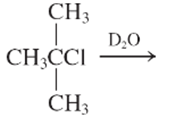
Concept Introduction:
A type of organic reaction in which two substituents get eliminated from a given molecule by one step mechanism or two step mechanism is known as elimination reaction. The reaction which occurs in one step is known as E2 reaction whereas the reaction which occurs in two steps is known as E1 reaction.
In E1 reaction, elimination of HX substituent takes place and produces a double bond. It is a unimolecular reaction.
(f)
Interpretation:
The E1 product for the following reaction should be written.
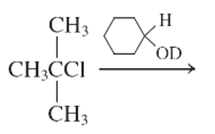
Concept Introduction:
A type of organic reaction in which two substituents get eliminated from a given molecule by one step mechanism or two step mechanism is known as elimination reaction. The reaction which occurs in one step is known as E2 reaction whereas the reaction which occurs in two steps is known as E1 reaction.
In E1 reaction, elimination of HX substituent takes place and produces a double bond. It is a unimolecular reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Organic Chemistry: Structure and Function
- Identify the correct transition state for an E2 reaction.arrow_forwardMake a table with the main conditions for each mechanism: E1, E2, SN1, SN2.arrow_forwardProvide the E2 mechanism for β-elimination reactionemploying 2-chloro-2-methylbutane to prepare 2-methyl-2-butene and 2-methyl-1-butene reaction. Use the actual structures of thereactants and products. Explain which of the alkenes is the major product ofthis reaction ?arrow_forward
- All rearrangements we have discussed so far have involved generation of an electron-deficient carbon followed by a 1,2-shift of an atom or a group of atoms from an adjacent atom to the electron-deficient carbon. Rearrangements by a 1,2-shift can also occur following the generation of an electron-deficient oxygen. Propose a mechanism for the acid-catalyzed rearrangement of cumene hydroperoxide to phenol and acetone.arrow_forwardComplete the following reactions, write major product(s) and specify the mechanism if ; SN1, Sn2, E, or E2 CH2Br + NaC=cCH;arrow_forwardFor each reaction indicate and explain the predominant mechanism: SN1, SN2, E1 or E2arrow_forward
- Provide a reaction mechanism and an appropriate set of conditions to carry out the reaction shown. он SN2 reaction Br2 in CH2C2 SN1 reaction KBr, DMSO SN1 meachanism HBr in H20 SN2 reaction KBr in DMSOarrow_forwardQ12. (1-bromoethyl)benzene 1 udergoes an elimination following an E1 mechanism. Fill in the following synthetic scheme by drawing the structure of intermediate 2 and product 3. The chemical formula of product 3 is provided as guidance. CH; RDS Br C3Hg E1 2 3arrow_forwardAnswer the question below the reaction. ta The reaction above proceeds through which type of mechanism? SN2 SN1 E1 E2 OH + Excess NH4C1 H₂SO4 + H₂Oarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

