
Organic Chemistry: Structure and Function
8th Edition
ISBN: 9781319079451
Author: K. Peter C. Vollhardt, Neil E. Schore
Publisher: W. H. Freeman
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 6, Problem 67P
Interpretation Introduction
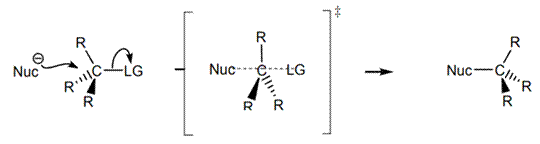
Interpretation: The ion among
Concept introduction: Bimolecular substitution or
A general
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Rank the following nucleophiles from weakest to strongest (e.g. least reactive to
most reactive).
1
O
OI<|| < |||
Please explain why this reaction will NOT WORK.
NO2
1. HNO3, H2SO4,
2. EtCI, AICI3
HO
NH2
CO₂Me
PG!
A
NH
HO
PG²
HO
CO₂Me
PG!
NH
`PG³
PG!
`NH
PG3
PG!
`NH
B
PG2
CO₂Me
C
PG20
LiBH4
strong reducing agent,
strong nucleophile
PG!
NH
OH
Chapter 6 Solutions
Organic Chemistry: Structure and Function
Ch. 6.2 - Prob. 6.1ECh. 6.2 - Prob. 6.3TIYCh. 6.3 - Prob. 6.4ECh. 6.3 - Prob. 6.5ECh. 6.3 - Prob. 6.6ECh. 6.3 - Prob. 6.7ECh. 6.3 - Prob. 6.8ECh. 6.4 - Prob. 6.10TIYCh. 6.4 - Prob. 6.11ECh. 6.5 - Prob. 6.12E
Ch. 6.5 - Prob. 6.13ECh. 6.6 - Prob. 6.14ECh. 6.6 - Prob. 6.16TIYCh. 6.6 - Prob. 6.17ECh. 6.7 - Prob. 6.18ECh. 6.7 - Prob. 6.19ECh. 6.7 - Prob. 6.20ECh. 6.8 - Prob. 6.21ECh. 6.8 - Prob. 6.22ECh. 6.8 - Prob. 6.23ECh. 6.8 - Prob. 6.24ECh. 6.9 - Prob. 6.26TIYCh. 6.10 - Prob. 6.27ECh. 6.10 - Prob. 6.28ECh. 6 - Prob. 31PCh. 6 - Prob. 32PCh. 6 - Prob. 33PCh. 6 - Prob. 34PCh. 6 - Prob. 35PCh. 6 - Prob. 36PCh. 6 - Prob. 37PCh. 6 - Prob. 38PCh. 6 - Prob. 39PCh. 6 - Prob. 40PCh. 6 - Prob. 41PCh. 6 - Prob. 42PCh. 6 - Prob. 43PCh. 6 - Prob. 44PCh. 6 - Prob. 45PCh. 6 - Prob. 46PCh. 6 - Prob. 47PCh. 6 - Prob. 48PCh. 6 - Prob. 49PCh. 6 - Prob. 50PCh. 6 - Prob. 51PCh. 6 - Prob. 52PCh. 6 - Prob. 53PCh. 6 - Prob. 54PCh. 6 - Prob. 55PCh. 6 - Prob. 56PCh. 6 - Prob. 57PCh. 6 - Prob. 58PCh. 6 - Prob. 59PCh. 6 - Prob. 60PCh. 6 - Prob. 61PCh. 6 - Prob. 62PCh. 6 - Prob. 63PCh. 6 - Prob. 64PCh. 6 - Prob. 65PCh. 6 - Prob. 66PCh. 6 - Prob. 67PCh. 6 - Prob. 68P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- reaction of C with OH in a different solvent has been shown to have a bimolecular slow step. Which nucleophile will react faster with OH (aq) A CH,0 15. CI CH,NH" В CH,CH, Select ALL that apply a d.arrow_forwardFigure 8-2 YOND H+ A. c. H30+ ET rxn + Je whi prog Ich Но веле ихл B. H₂O: un гхи prog P. E1 tr ГХИ 13 prog 3 H₂ő:arrow_forward4. Predict the products of each of the following reactions, and indicate the nucleophile that is formed. so 2. NaOH, Br₂ 1. CH,CH,NHCH,CH pH 5 3. H₂O+ H₂O+, Cl₂arrow_forward
- Draw an energy diagram of the following reactions. A one step reaction with a positive AG A one step reaction with a negative AG A two-step reaction with an overall positivel-Where the intermediates are lower energy than the reactants and the first transition state is lower in energy than the second.arrow_forwardThe following figure shows the energy profile of a nucleophilic substitution reaction. Potential energy :ÖH H HII.C H C-1 H HO--C---I HH Reaction coordinate H -CH HO–CHITH H + 1 According to this, it is correct to affirm: O The activation energy (4G°) shows that it is thermodynamically favorable. O Represents a reaction that occurs in one step where a pentavalent chemical species is formed. O The intermediate has lower energy than any transition state. O The formation of the intermediate releases energy and is therefore thermodynamically favourable.arrow_forward7. Reaction Scheme. NH₂ NH2 or two differnet methods (no same steps/reagents) C5H12N2 1. xs Mel, xs K2CO3 2. Ag2O, H₂O 3. heat Br2, xs NaOH, xs H₂O OHC 1.03 2. DMS CHOarrow_forward
- Pls help ASAP. Pls do all asked questions. Pls identify the reaction below.arrow_forwardWhich chlorination product is formed via the lowest energy transition state in the rate-determining step? + CI-CI de .CI O All radical chlorinations require the same activation energies.arrow_forwardIn a reaction sequence, C has a lower free energy than A, and k2 » k_,» kj. Draw an energy profile and derive the kinetic equation I k₁ A k B- k₂ Carrow_forward
- OH + H* Under what condition would the reaction proceed faster? O A. In the presence of a salt O B. Acid-base mixture catalyzed O. Base catalyzed O D. Acid catalyzedarrow_forwardWhich of the following species are not nucleophiles: Na+, Cl-, +CH3, :NH3, HO:-?arrow_forwardIt is not necessary for a nucleophile to have an unshared electron pair. O True O Falsearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License