
Concept explainers
(a)
Interpretation: Nucleophile that is better suited for its role in
Concept introduction: Bimolecular substitution or
A general
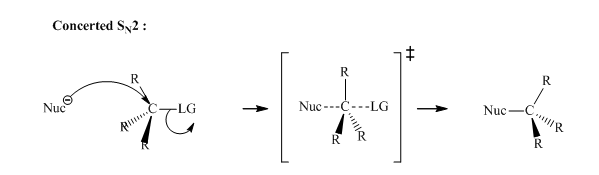
Polar-aprotic solvents accelerate the rate of
(b)
Interpretation: Substrate that is better suited for its role in
Concept introduction: Bimolecular substitution or
A general
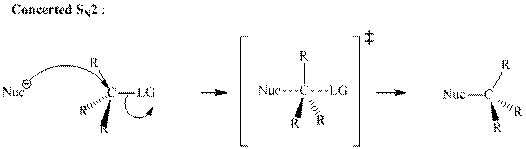
Polar-aprotic solvents accelerate the rate of
(c)
Interpretation: Solvent that is better suited for its role in
Concept introduction: Bimolecular substitution or
(d)
Interpretation: Leaving group that is better suited for its role in
Concept introduction: Bimolecular substitution or
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry: Structure and Function
- Write the products of the reactions below and show the mechanism by which they are formed. но catalytic HCl water/THF solvent Ph conc. H,SO4arrow_forwardConsider the attached SN2 reaction. Question: What happens to the reaction rate in each of the following instances? [1] The leaving group is changed from Br− to I−; [2] The solvent is changed from acetone to CH3CH2OH; [3] The alkyl halide is changed from CH3(CH2)4Br to CH3CH2CH2CH(Br)CH3; [4] The concentration of −CN is increased by a factor of five; and [5] The concentrations of both the alkyl halide and −CN are increased by a factor of fivearrow_forward8) Write the mechanism of the following reaction? 1. NABH4 2. H20 OHarrow_forward
- Convert 2-pentanol into 2,3-dibromopentane. Draw structures of the starting material (2-pentanol) and final product (2,3-dibromopentane), and show the two reactions needed for this synthesis. Include the structure of the intermediate compound, and the reagents and conditions for each reaction. Then explain why 1,2-dibromopentane would not be a significant product of this synthesis.arrow_forwardFor the following reaction there are three (3) possible products. Two of the products are expected to be minor and one product is expected to be a major product. Br H₂O Name and draw out the step-by-step mechanisms by which each of the products is formed. Briefly explain why one of the three products is expected to be a major product.arrow_forwardIn both examples below the reactants shown are combined to bring about a nucleophilic substitution (Sn1, Sn2) and/or elimination (E1, E2) reaction. What is the major reaction that takes place in each case? CH2CI NaOH H2O CH3 CH3 CH,CH,CHCCH, H20arrow_forward
- Each of the following molecules has two leaving groups (LG) at different positions. If 0.1M each molecule is reacted with 0.1 M OH in an SN2 reaction, indicate which LG in each molecule will be more reactive and why. Show the products of these reactions. osO,Me Br Br oso,CF3arrow_forwardWrite a mechanism for the following reaction. AIBN = AIBN, heat .N. CN NC CH,SH 2.arrow_forward1a) What will be the product of the following reaction? O₂N- Br + (HO)₂B Pd(OAc)₂ Degassed H₂O/Acetonearrow_forward
- Consider the compound below, with all its carbons labeled. In reality we might predict that the alkene groups are more reactive to electrophiles, but instead imagine that an electrophile binds to carbon five as the first step of an electrophilic aromatic substitution mechanism. A carbocation results, with multiple resonance structures. Select every carbon that bears a positive charge in one of these resonance structures. 10 Carbon 1 O Carbon 2 Carbon 3 OCarbon 4 OCarbon 5 OCarbon 6 Carbon 7 Carbon 8 Carbon 9 Oerboi 10.arrow_forwardGive the products of the following substitution reactions. For every reaction, show electron pairs on both nucleophile and leaving group.arrow_forwardIn both examples below the reactants shown are combined to bring about a nucleophilic substitution (SN1, SN2) and/or elimination (E1, E2) reaction. What is the major reaction that takes place in each case? -Br CH3 CH3CH₂CCH3 ci Nal acetone NaOCH₂CH3 CH3CH₂OH SN2 SN2arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

