
Concept explainers
a.
Interpretation: The mechanism using curved-arrow notation for the following reaction needs to be determined if reaction is possible:
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as
b.
Interpretation: The mechanism using curved-arrow notation for the following reaction needs to be determined if reaction is possible:
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
c.
Interpretation: The mechanism using curved-arrow notation for the following reaction needs to be determined if reaction is possible:
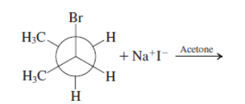
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
d.
Interpretation: The mechanism using curved-arrow notation for the following reaction needs to be determined if reaction is possible:
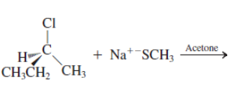
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
e.
Interpretation: The mechanism using curved-arrow notation for the following reaction needs to be determined if reaction is possible:
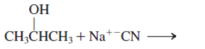
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
f.
Interpretation: The mechanism using curved-arrow notation for the following reaction needs to be determined if reaction is possible:
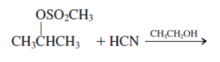
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
g.
Interpretation: The mechanism using curved-arrow notation for the following reaction needs to be determined if reaction is possible:
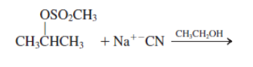
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
h.
Interpretation: The mechanism using curved-arrow notation for the following reaction needs to be determined if reaction is possible:
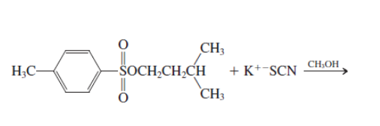
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
i.
Interpretation: The product for the following reaction needs to be determined if reaction is possible:
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
j.
Interpretation: The product for the following reaction needs to be determined if reaction is possible:
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry: Structure and Function
- Provide the missing reagent, intermediate, transition state or major product for these reactionsarrow_forwardFor the reaction can someone please synthesize the given products from the given reactants. Multiple reactions/steps will be needed (the arrows designate the minimum number of steps). Also, for the 1st step (reaction) in each synthesis, can you please draw an energy diagram showing the correct number of hills and valleys for that step’s mechanism?arrow_forwardSelect the reactions in Figure 39 that can be represented by the accompanying energy diagram?arrow_forward
- For each of the steps in Problem 7.26, determine whether the product can eliminate a leaving group to produce a compound that is different from the reactants. For those that can, draw the appropriate curved arrows and the new product that forms.arrow_forwardQ1. Draw the reaction mechanism using curly arrows to show each bond made/broken in the production of biodiesel from vegetable oil. Q2. NaOH is in a catalytic amount, why is it not needed in equimolar amount to the Methanol?arrow_forwardConsider the following overall reaction, which will be discussed in Chapter 20.arrow_forward
- Fill in the blanks: Progress of the reaction This is the reaction diagram of reactant A converted to product D (A → D). The reaction has 2 intermediates. It has 3 transition states. The fastest step in the reaction is the conversion of c to d because it has the lowest activat Is the first step of the reaction (exergonic or endergonic?) because is The overall reaction is (exergonic or endergonic?) because Free energyarrow_forwardI was able to do a few of these on my own, but I'm really stuck with this one. Should I be flipping the third reaction? If so, how does that help me cancel out the reactants and products on both sides? Which ones should I be multiplying?arrow_forwardWhat major product (from Figure #1) results from the following dehydration reaction (from Reaction #1)? Reaction #1 HO, H2SO4 heat Figure #1 compound A compound B compound C compound D Please click here if image does not display. In predicting the major product of boxed reaction #1, it is strongly recommended that you work out a complete mechanism on a piece of scratch paper. compound C compound A compound D compound Barrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





