
Concept explainers
(a)
Interpretation:The product of the reaction of
Concept introduction: Bimolecular substitution or
A general
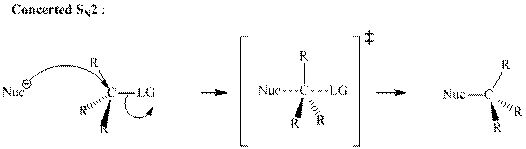
A nucleophile is an electron-rich species with high electron density that can be donated to another electron-deficient species called electrophile. For effective
(b)
Interpretation: The product of the reaction of
Concept introduction:Bimolecular substitution or
A general
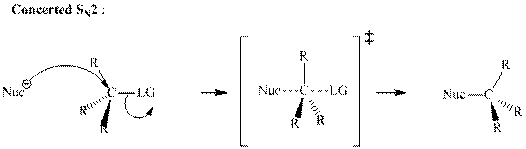
A nucleophile is an electron-rich species with high electron density that can be donated to another electron-deficient species called electrophile. For effective
(c)
Interpretation: The product of the reaction of
Concept introduction:Bimolecular substitution or
A general
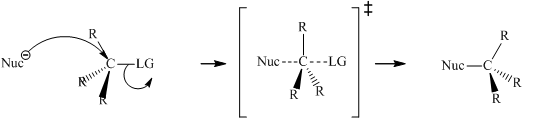
A nucleophile is an electron-rich species with high electron density that can be donated to another electron-deficient species called electrophile. For effective
(d)
Interpretation: The product of the reaction of
Concept introduction:Bimolecular substitution or
A general
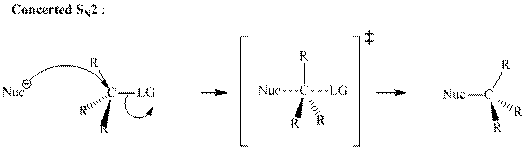
A nucleophile is an electron-rich species with high electron density that can be donated to another electron-deficient species called electrophile. For effective
(e)
Interpretation: The product of the reaction of
Concept introduction:Bimolecular substitution or
A general
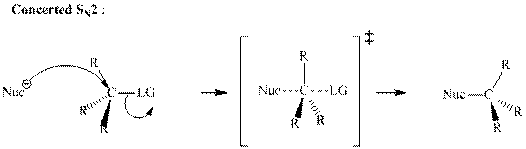
A nucleophile is an electron-rich species with high electron density that can be donated to another electron-deficient species called electrophile. For effective
(f)
Interpretation: The product of the reaction of
Concept introduction:Bimolecular substitution or
A general
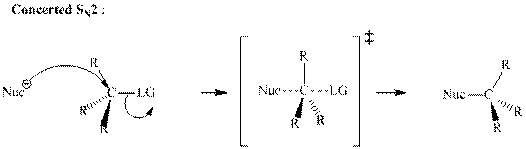
A nucleophile is an electron-rich species with high electron density that can be donated to another electron-deficient species called electrophile. For effective
(g)
Interpretation: The product of the reaction of
Concept introduction:Bimolecular substitution or
A general
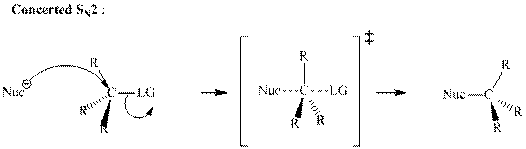
A nucleophile is an electron-rich species with high electron density that can be donated to another electron-deficient species called electrophile. For effective
(h)
Interpretation: The product of the reaction of
Concept introduction:Bimolecular substitution or
A general
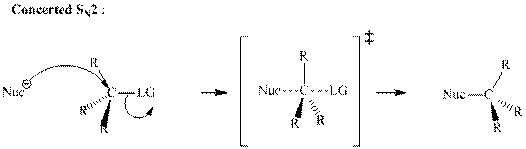
A nucleophile is an electron-rich species with high electron density that can be donated to another electron-deficient species called electrophile. For effective
(i)
Interpretation: The product of the reaction of
Concept introduction:Bimolecular substitution or
A general
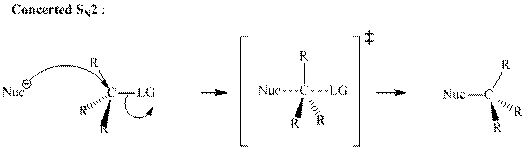
A nucleophile is an electron-rich species with high electron density that can be donated to another electron-deficient species called electrophile. For effective
(j)
Interpretation: The product of the reaction of
Concept introduction:Bimolecular substitution or
A general
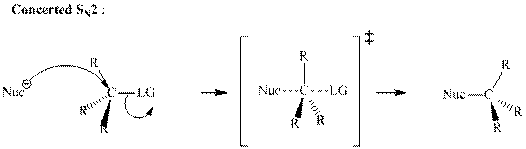
A nucleophile is an electron-rich species with high electron density that can be donated to another electron-deficient species called electrophile. For effective
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry: Structure and Function
- Show reagents and experimental conditions to synthesize the following compounds from 1-propanol (any derivative of 1-propanol prepared in one part of this problem may be used for the synthesis of another part of the problem). (a) Propanal (b) Propanoic acid (c) Propene (d) 2-Propanol (e) 2-Bromopropane (f) 1-Chloropropane (g) 1,2-Dibromopropane (h) Propyne (i) 2-Propanone (j) 1-Chloro-2-propanol (k) Methyloxirane (l) Dipropyl ether (m) Isopropyl propyl ether (n) 1-Mercapto-2-propanol (o) 1-Amino-2-propanol (p) 1,2-Propanediolarrow_forwardWhen 2-pentene is treated with Cl2 in methanol, three products are formed. Account for the formation of each product (you need not explain their relative percentages).arrow_forwardFollowing is a balanced equation for the allylic bromination of propene. CH2==CHCH3 + Br2 h CH2==CHCH2Br + HBr (a) Calculate the heat of reaction, H 0, for this conversion. (b) Propose a pair of chain propagation steps and show that they add up to the observed stoichiometry. (c) Calculate the H 0 for each chain propagation step and show that they add up to the observed H 0 for the overall reaction.arrow_forward
- Select the reagent that you would use to convert an alkene to a Markovnikov (more substituted) alcohol without possibility of carbocation rearrangement. o H2SO4, H2O OBH3-THF, then H2O2, H2O, NaOH Hg(OAc)2, H2O, then NaBH4 o OsO4, TBHP, H2Oarrow_forwardWrite an equation for the acid-base reaction between 2,4-pentanedione and sodium eth- oxide and calculate its equilibrium constant, K. The pK, of 2,4-pentanedione is 9; that of ethanol is 15.9. CH,CCHÖCH, + CH,CH,O Na* H 2,4-Pentanedione Sodium ethoxidearrow_forwardEthers can be prepared by reaction of an alkoxide or phenoxide ion with a primary alkyl halide. Draw the structure of the expected organic product of the reaction of iodomethane with the following alkoxide ion: CH3 H3C O Na You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Do not include counter-ions, e.g., Na", I, in your answer. орy вste ChemDoodlearrow_forward
- Starting with 1-butanol, select the reagents you would use to prepare the following compound: OH LiAlH4 followed by H3O+ Na2Cr₂O7, H₂SO4, H₂O NaBH4, MeOH O PCC, CH₂Cl₂arrow_forward10) Synthesis: Make the following products from a suitable cyclic alkene starting material. Look at the functional group PATTERN present in the molecule, including stereochemistry. Br CH3 Br CH3arrow_forwardEthers can be prepared by reaction of an alkoxide or phenoxide ion with a primary alkyl halide. Draw the structure of the expected organic product of the reaction of 1-iodo-2-methylpropane with the following alkoxide ic + O Na You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Do not include counter-ions, e.g., Na", I", in your answer. C opy astearrow_forward
- Draw the line-angle formula of the enol formed in the following alkyne hydration reaction and then draw the structural formula of the carbonyl compound with which this enol is in equilibrium. -C=CH 1. (sia)2BH 2. NaOH/H₂O₂ an enol carbonyl compound • You do not have to consider stereochemistry. • Draw both the enol and the carbonyl forms. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Separate structures using the sign from the drop-down menu. ChemDoodleⓇ <arrow_forwardlist the reagents that are needed to carry out in these reactionsarrow_forwardThe reaction shown below is carried out in two stages. In the first, the two reactants are combined in tetrahydrofuran (THF) solvent to form a neutral adduct. Aqueous acid is then added to hydrolyze this initial adduct, giving the final product. N(CH3)2 I. H a For the reaction shown above, draw the major organic product having at least one carbonyl group. • You do not have to consider stereochemistry. Pis 85 1. THF 2. H3O+ n [ ]#arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
