
Concept explainers
Interpretation: The synthesis process of given molecule from methylcyclohexane needs to be explained.

Concept Introduction: A
In a chemical reaction; the substance which is involved in conversion is said to be reactant whereas the newly formed substance is called as a product. Both reactant and products must be separated by an arrow.
Halogenation reaction is an addition reaction in which the halogen atoms like Cl, Br are bonded on un-statured carbon atoms of
Interpretation: The synthesis process of given molecule from methylcyclohexane needs to be explained.
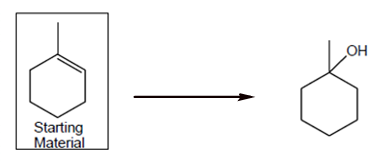
Concept Introduction: A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction; the substance which is involved in conversion is said to be reactant whereas the newly formed substance is called as a product. Both reactant and products must be separated by an arrow.
Halogenation reaction is an addition reaction in which the halogen atoms like Cl, Br are bonded on un-statured carbon atoms of alkene to form alkyl halide.
Interpretation: The synthesis process of given molecule from methylcyclohexane needs to be explained.
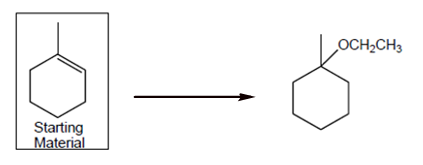
Concept Introduction: A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction; the substance which is involved in conversion is said to be reactant whereas the newly formed substance is called as a product. Both reactant and products must be separated by an arrow.
Halogenation reaction is an addition reaction in which the halogen atoms like Cl, Br are bonded on un-statured carbon atoms of alkene to form alkyl halide.
Interpretation: The synthesis process of given molecule from methylcyclohexane needs to be explained.

Concept Introduction: A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction; the substance which is involved in conversion is said to be reactant whereas the newly formed substance is called as a product. Both reactant and products must be separated by an arrow.
Halogenation reaction is an addition reaction in which the halogen atoms like Cl, Br are bonded on un-statured carbon atoms of alkene to form alkyl halide.
Interpretation: The synthesis process of given molecule from methylcyclohexane needs to be explained.

Concept Introduction: A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction; the substance which is involved in conversion is said to be reactant whereas the newly formed substance is called as a product. Both reactant and products must be separated by an arrow.
Halogenation reaction is an addition reaction in which the halogen atoms like Cl, Br are bonded on un-statured carbon atoms of alkene to form alkyl halide.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Organic Chemistry: A Guided Inquiry
- Predict the initial and final products for the following reaction involving 2‑pentyne or pent‑2‑yne. Hint: the products are isomers and in equilibrium with each other.arrow_forwardWhich alkene product would you expect to be the major product under kinetic conditions? Under thermodynamic conditions? The given pictures are the reactions in which we have to determine which alkene product we expect would be the major product under kinetic and thermodynamic conditions.arrow_forwardWhat alkene should be used to synthesize each of the following alkyl halides?arrow_forward
- a) How many different molecules containing one double bond can be used to synthesize 4-methyl-4-heptanol (as the major product) via acid catalyzed hydration with H2SO4 and H2O? Draw them all below. b) Are any of the structures in part (a) trisubstituted alkenes? If so, redraw it/them below.arrow_forward[References] Draw the alkene structure that produced the following compounds in a ozonolysis reaction as specified. 1. O3 C7H12 2. (CH3)2Sarrow_forwardThe following reaction could produce an isomeric mixture of three alkenes. Please answer the following questions.arrow_forward
- 2) Provide the structure of the major organic product of the following reaction. O HN C₂H₂COC1 AlCl3arrow_forwardBased on the images attached, choose the alkene product formed for the following reaction and predict the major and minor compounds.arrow_forward5. Synthesis: Synthesize the following compound from cyclohexane, any carbon pieces that are three carbons or less (can include other functional groups), and any inorganic reagents.arrow_forward
- In the reaction (E) 2-methyl -2,4-hexadiene with hydrogen bromide at room temperature, two isomeric products are isolated. Draw the structures for these isomeric products. Which is the kinetic isomer and which is the thermodynamic isomer.arrow_forwardRank the following aldehydes and ketones in order from LEAST reactive to MOST reactive and explain why.arrow_forwardExplain why the following alcohols, when heated with acid, form the same alkene.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





