
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 8, Problem 11E
Interpretation Introduction
Interpretation: Structure for the product and a reasonable mechanism for below reaction should be drawn.
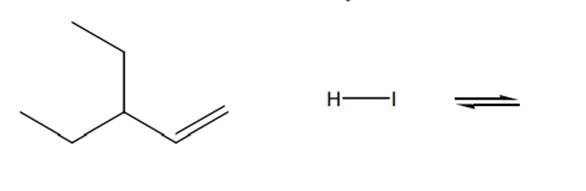
Concept introduction:
The product formed is governed by Markovnikov’s Rule. Rule suggests that negative part of halo acid HX must go to the carbon that has more alkyl substituents or less
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
pls give me answer of this question with explanation immiditely and i will rate you sir.
Looking at the NMR, how can you determine the compound is cyclohexane carboxaldehyde?
Distinguish all the ppm (be detailed)
Which structure is in agraement with both of thasa R and H NMA data?
IR, cm (Intensity)-2965 (strong), 1745 (strong), 1238 (strong)
H NMR, ppm (splitting, H): 0.94 (doublet, 6H), 193 (multiplet, 1H), 2.05 (singlet, 3H), 3.8as (doublet,
2H)
A.
tor
B.
D.
C.
Chapter 8 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 8 - Prob. 1CTQCh. 8 - Prob. 2CTQCh. 8 - Prob. 3CTQCh. 8 - Prob. 4CTQCh. 8 - Prob. 5CTQCh. 8 - Prob. 6CTQCh. 8 - Prob. 7CTQCh. 8 - Prob. 8CTQCh. 8 - Prob. 9CTQCh. 8 - Prob. 10CTQ
Ch. 8 - Draw the products that result from the electron...Ch. 8 - Prob. 12CTQCh. 8 - Draw the products that would result if the arrow...Ch. 8 - Prob. 14CTQCh. 8 - What information (if any) from the following...Ch. 8 - Prob. 16CTQCh. 8 - Prob. 17CTQCh. 8 - The reactants, intermediates, final products, and...Ch. 8 - Prob. 19CTQCh. 8 - Prob. 20CTQCh. 8 - Prob. 21CTQCh. 8 - Prob. 22CTQCh. 8 - Explain how you can tell from the energy diagram...Ch. 8 - Explain why the following mechanism for hydration...Ch. 8 - Prob. 25CTQCh. 8 - Prob. 26CTQCh. 8 - Prob. 27CTQCh. 8 - Prob. 28CTQCh. 8 - Prob. 29CTQCh. 8 - Prob. 30CTQCh. 8 - Prob. 31CTQCh. 8 - The hydration above is one of a family of...Ch. 8 - Prob. 33CTQCh. 8 - Which statement is false? a. A mechanistic step...Ch. 8 - Prob. 35CTQCh. 8 - Prob. 36CTQCh. 8 - Prob. 37CTQCh. 8 - Draw the complete mechanism including the...Ch. 8 - Prob. 2ECh. 8 - Explain why ethene does not react with HX ( X=Cl ,...Ch. 8 - Draw the complete mechanism of each pair of...Ch. 8 - Prob. 5ECh. 8 - Prob. 6ECh. 8 - Prob. 7ECh. 8 - Prob. 8ECh. 8 - Prob. 9ECh. 8 - Prob. 10ECh. 8 - Prob. 11ECh. 8 - Prob. 12ECh. 8 - Prob. 15ECh. 8 - A student proposes the following reaction...Ch. 8 - Prob. 17ECh. 8 - Prob. 18ECh. 8 - Prob. 19E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Why the extended Pi(π) conjugated are suitable for Fluorescence properties? Please shortly answer at your own words. Answer should be to the point(specific 4-5 lines).arrow_forwardPredict the structure of the compound with the following spectraarrow_forwardProvide the complete mechanism using CAF for the reaction shown below. Tell how many signals are visualized in the 1H spectrum for the starting material and the product. H₂O, H₂SO4 gr=gr NH₂ heat OH + NH4+ HSO4arrow_forward
- Determine the structure of the compound using the information provided below, show the proton integration and the mass spectrometry fragments (ensure curly arrow mechanism).arrow_forwardI need help reading these NMR. I need to identify the solven peak for each, and I need to differentiate the peaks between brominated and debrominated. I would appreciate an explanation, thank you!arrow_forwardBased on MS spectrum and the compound given, provide mechanistic explanations for fragmentations in the MS spectrum.arrow_forward
- Looking at the NMR & Mass Spectrum, how do you determine the compound is "cyclohexane carboxaldehyde"?arrow_forwardDeaw the structure of the compound whose Sper spectroscopy data speef provided, сонго 2.6 ppm, 1H! sextet 2.1ppm, 34; singlet 1.7 -1.3 ppm, 2H, multipet 1.1 ppm, 3H, doublet 0.9 ppm, 34; triplet 3H iarrow_forwardThe following H-NMR spectrum (showing all available signals) is for one of the possible products listed below. This product was obtained from oxidizing one of the alcohols listed below (as possible reactants) using either dichromate (Na2Cr2O7/H2SO4) or DMP. Determine the product, reactant and oxidizing agent following the analysis below. Based on the given H-NMR spectrum, and since there is signals is and the total number of H-NMR the oxidation product must be ÷ Therefore, the oxidizing agent and the reactant must be PPM Possible Reactants OH A Possible Products O I V ОН B ОН VI II ОН C ОН OH III VII OH D OH E IV VIIIarrow_forward
- please label the corresponding CNMR and HNMR spectra peaks on this molecules.arrow_forwardThe product from the following reaction shows infrared absorptions at 3050 cm', 2930 cm', 1600 cm', 1570 cm', 1490 cm', 1450 cm", and 1150 cm', and a broad absorption around 3550 cm structure of the product. Use this spectroscopic data and the appended infrared correlation table to make clear assignments of all the listed absorptions to explain your reasoning. Draw the a.) LİAIH4 OCH3 b.) dil. aq. HCI infrared assignments: absorption (cm') assignment 3550 3050 2930 1600 1570 1490 1450 1150arrow_forwardAn oxidation reaction was conducted using cyclohexanol and NaOCl to make cyclohexanone. How can we know that the NMR for cyclohexanone is in fact cyclohexanone( the NMR for cyclohexanone is not labeled). Analyze the spectra to prove its identity along with the NMR of the starting material which is the spectra labeled cyclohexanol below.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License