
(a)
Interpretation: The mechanism for formation of the alcohol product should be drawn.
Concept introduction:
The product formed is governed by Markovnikov’s Rule. Rule suggests that negative part of halo acid HX must go to the carbon that has more alkyl substituents or less
The order of relative stability of various possible carbocation species is as follows:
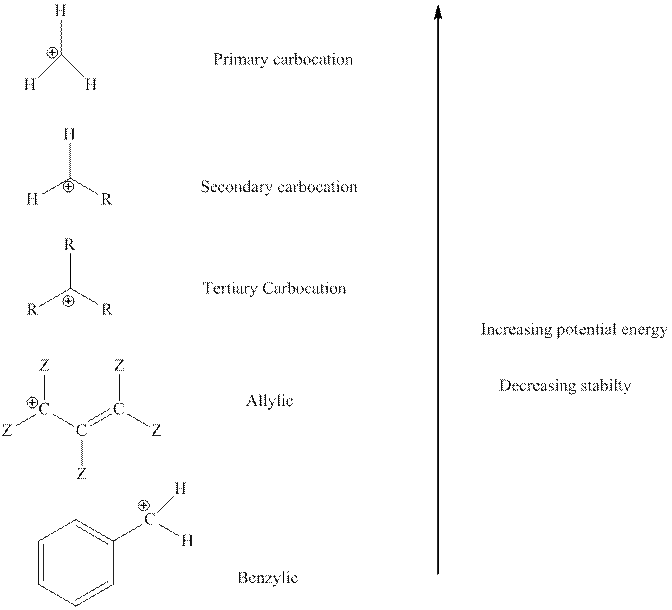
(b)
Interpretation: The expected product of reaction was carried out in methanol instead of water should be drawn.
Concept introduction: Alkenes are regarded electron-rich and often undergo addition reaction in presence of electrophilic halo acids or nucleophilic sodium cyanide. The intermediate formed in former is tertiary carbocation and finally nucleophilic part of halo acid attacks in a rapid step to give addition product.
The product formed is governed by Markovnikov’s Rule. Rule suggests that negative part of halo acid HX must go to the carbon that has more alkyl substituents or less
The order of relative stability of various possible carbocation species is as follows:
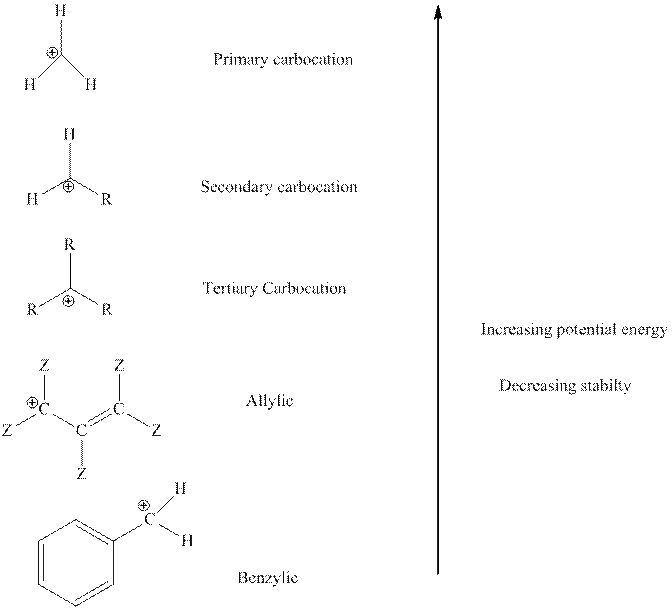
Trending nowThis is a popular solution!

Chapter 8 Solutions
Organic Chemistry: A Guided Inquiry
- 1. Attack the alkyl chloride with the stronger nucleophile. 2. Deprotonated to form the productarrow_forward7. Complete the following reaction and propose a mechanism.arrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts and the alcohol side product. 1. LIOH 2. Neutralizing work-up Select to Drawarrow_forward
- HS HCI a. b. c. Draw a curly arrow mechanism for this reaction to explain the formation of each product. Your mechanism should be consistent with your responses to parts a and b. Does this reaction proceed via an SN1 or an SN2 reaction? Explain your choice. Redraw each reactant and include the orbitals that interact during this substitution reaction. Label each orbital as either the HOMO or LUMO.arrow_forward6. Multi Step Synthesis. Propose a synthetic route from the reactant to the product using any reagents you need. Nalt LINH4arrow_forward1. Draw the products of each nucleophilic substitution reaction a. b. D b OH C. d. e. f. Br 1 NaCN + NaOCH3 H₂Oarrow_forward
- For each reaction, write the letter corresponding to the type of reaction (before the number) and draw the MAJOR product (in the product side). A. Nucleophilic Addition B. Nucleophilic Condensation C. Nucleophilic Acyl Substitutionarrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts and the alcohol side product. 1. excess LIАІН4 2. Н2О P Type here to searcharrow_forward9. Under normal conditions, 1-cyclohexenol cannot be isolated or stored in a bottle. a. Explain why 1-cyclohexenol cannot be isolated or stored. b. Draw the product that would be isolated and stored instead of 1-cyclohexanol. c. Draw a mechanism to explain your answer above.arrow_forward
- 6. Circle the structure with the most nucleophilic double bond. Put a box around the structure with the least nucleophilic double bond. OHarrow_forwardFor alkylhalides, elimination reaction is much favored than substitution reaction when A. Temperature is relatively high. B. Temperature is relatively low. C. Base is relatively weak D. Pressure is relatively high.arrow_forwardDraw the final products A and B for the following reactions.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
