
Concept explainers
Interpretation: Reason for rearrangement shown below shows downhill in terms of potential energy should be explained.
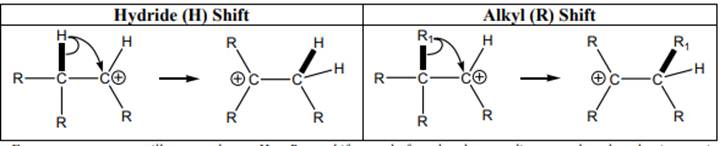
Concept introduction: Carbocation is a general term employed for a postively charged carbon irrespective of valency of carbon. In carbocation, carbon is bonded to 3 atoms or groups and has only sextet of electrons so it behaves as an electron-deficient species. It is
The carbocation formed may rearrange itself to a more stable carbocation through a
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Organic Chemistry: A Guided Inquiry
- Draw mechanism for the reactions below. Make sure to show all "electron-pushing" arrows, cond and lone pairs.arrow_forwardpts) Show any reaction that would involve the rearrangement of an intermediate carbocation.arrow_forwardOCHEM help... What is the major product of the following reaction sequence? (See attached image)arrow_forward
- Draw both resonance structures of the most stable carbocation intermediate in the reaction shown. Please circle the one I need to draw in the space below.arrow_forwardDraw the product by following the curved arrows. This reaction is an example of a [3,3] sigmatropic rearrangement,arrow_forward함 H Follow the curved arrows and draw the product of this reaction. . You do not have to consider stereochemistry.arrow_forward
- Brarrow_forwardThis content is protected and may nag shared t 4. Consider the following reaction. This reaction is a multi-step reaction. [1] Draw the mechanism for each step of this reaction using curved arrow notation, include all lone pairs of electrons if they are directly involved in the reaction. [2] Identify the arrow pushing pattern (mechanistic pattern: nucleophilic attack, loss of leaving group, or proton transfer) for each step. [3] Draw a transition state for each step. H-OSO3H CH3 00 H H3C-O-H Хосно H3C-O-H Harrow_forwarda) Show a detailed mechanism for the reaction to produce nitrogen gas and the two carbon radicals shown above.b) Show how the carbon radical produced above could react with HBr to make a bromine radical, completing the initiation step for the reaction at the top of the page.c) Show a detailed mechanism for the propagation step of the reaction at the top of the page and circle the major organic product produced from the reaction.arrow_forward
- Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron- pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. Probler Drawing Arrows THF Na :Cl:O 'N. NaO C:O Drag Toarrow_forwardDraw the missing produts for the reaction shown below. Be sure to include stereochemistry where appropriate.arrow_forwardPlease use appropriate reagent to transform the following . GIVE REASON FOR USING A PARTICULAR REAGENT AND MUST GIVE MECHANISM PLZ . DO NOT SKIP MECHANISM. DO ALL , PLZ DO NOT COPY FROM CHEGG if you plan to do only 1 then skip lets some other do allarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
