
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 8, Problem 33CTQ
Interpretation Introduction
Interpretation: Number of synthetic steps below reaction should be stated.
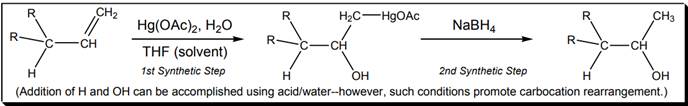
Concept introduction: The carbon-carbon double bond in
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Show work. don't give Ai generated solution
Briefly explain the existence of Nb-Nb bond in the alpha-NbI4 compound.
In the case of isopilianions, briefly state:- why polymeric species with a defined MW are formed.- why the extent of polymerization is different depending on the metal.- why these polyhedra with such special structures are formed.
Chapter 8 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 8 - Prob. 1CTQCh. 8 - Prob. 2CTQCh. 8 - Prob. 3CTQCh. 8 - Prob. 4CTQCh. 8 - Prob. 5CTQCh. 8 - Prob. 6CTQCh. 8 - Prob. 7CTQCh. 8 - Prob. 8CTQCh. 8 - Prob. 9CTQCh. 8 - Prob. 10CTQ
Ch. 8 - Draw the products that result from the electron...Ch. 8 - Prob. 12CTQCh. 8 - Draw the products that would result if the arrow...Ch. 8 - Prob. 14CTQCh. 8 - What information (if any) from the following...Ch. 8 - Prob. 16CTQCh. 8 - Prob. 17CTQCh. 8 - The reactants, intermediates, final products, and...Ch. 8 - Prob. 19CTQCh. 8 - Prob. 20CTQCh. 8 - Prob. 21CTQCh. 8 - Prob. 22CTQCh. 8 - Explain how you can tell from the energy diagram...Ch. 8 - Explain why the following mechanism for hydration...Ch. 8 - Prob. 25CTQCh. 8 - Prob. 26CTQCh. 8 - Prob. 27CTQCh. 8 - Prob. 28CTQCh. 8 - Prob. 29CTQCh. 8 - Prob. 30CTQCh. 8 - Prob. 31CTQCh. 8 - The hydration above is one of a family of...Ch. 8 - Prob. 33CTQCh. 8 - Which statement is false? a. A mechanistic step...Ch. 8 - Prob. 35CTQCh. 8 - Prob. 36CTQCh. 8 - Prob. 37CTQCh. 8 - Draw the complete mechanism including the...Ch. 8 - Prob. 2ECh. 8 - Explain why ethene does not react with HX ( X=Cl ,...Ch. 8 - Draw the complete mechanism of each pair of...Ch. 8 - Prob. 5ECh. 8 - Prob. 6ECh. 8 - Prob. 7ECh. 8 - Prob. 8ECh. 8 - Prob. 9ECh. 8 - Prob. 10ECh. 8 - Prob. 11ECh. 8 - Prob. 12ECh. 8 - Prob. 15ECh. 8 - A student proposes the following reaction...Ch. 8 - Prob. 17ECh. 8 - Prob. 18ECh. 8 - Prob. 19E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A carboxylic acid reacts with water to form a carboxylate ion and H,O+. Complete the reaction. reaction: (CH),CHCH2COOH + H2O (CH), CHCH, COO¯ + H₂O+ Write the IUPAC name of the carboxylate ion formed in the reaction. IUPAC name: BIU X2 SPECIAL GREEK ALPHABET ~ Iarrow_forwardShow work. Don't give Ai generated solutionarrow_forwardA solution contains 10-3 M (NH4)2CO3 plus 10-3 M CaCO3. (NH4+: pKa 9.26) a) Follow the four steps and list the species and equations that would have to be solved to determine the equilibrium solution composition. (15 pts) b) Prepare a log C-pH diagram for the solution. Use a full sheet of graph paper, and show the ranges 1≤ pH < 13 and -10≤ log C≤ -1. (10 pts) c) Use the graphical approach for the solution pH. What is the concentration of all species? (15 pts)arrow_forward
- Keggin structure.arrow_forwardGiven: N2(g) + 3H2(g)2NH3(g) AG° = 53.8 kJ at 700K. Calculate AG for the above reaction at 700K if the reaction mixture consists of 20.0 atm of N2(g), 30.0 atm of H2(g), and 0.500 atm of NH3(g). A) -26.9 kJ B) 31.1 kJ C) -15.6 kJ D) 26.9 kJ E) -25.5 kJarrow_forwardExplain the structure of the phosphomolybdate anion [PMo12O40]3-.arrow_forward
- Keggin structure of heteropolyanions.arrow_forwardPlease correct answer and don't used hand raitingarrow_forwardAt pil below about 35 woon (Fe) oxidizes in streams according to the following Water in a reservoir at 20°C has a pH of 7.7 and contains the following constituents: Constituent (g) + Conc. (mg/L) Ca2+ 38 HCO3 abiotic oxid 183 HO Ferrous iron under these conditions and at 20°Cis Estimate the activities of Ca2+ and HCO3-, using an appropriate equation to compute the activity coefficients. (atomic weight: Ca 40)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning