
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
6th Edition
ISBN: 9781305687875
Author: Gilbert
Publisher: CENGAGE LEARNING - CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Question
Chapter 7.3, Problem 7E
Interpretation Introduction
Interpretation:
Whether the given isomers are diastereoisomers or enantiomers needs to be determined.
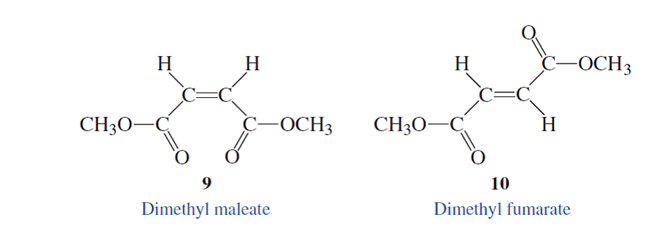
Concept Introduction :
The stereoisomers having different configurations at all stereocenters are known as enantiomers of each other and if there is a different configuration at only one stereocenter but same at other and both isomers are a non-mirror image of each other, and they will be diastereomers.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the structure's enantiomer. What are the structure's absolute configuration and its enantiomer's configuration?
How many chiral centers are in each of the molecules and how to find them?
Define the relationship between the molecules ( enantiomers , diastereomers structural isomers ( constitutional isomers ) , or identical . Locate the chiral centers and assign the absolute configuration for each of them .
Chapter 7 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
Ch. 7.2 - Prob. 1ECh. 7.2 - Prob. 2ECh. 7.2 - Prob. 3ECh. 7.2 - Prob. 4ECh. 7.2 - Prob. 5ECh. 7.2 - Prob. 6ECh. 7.2 - Prob. 7ECh. 7.2 - Prob. 8ECh. 7.2 - Prob. 9ECh. 7.2 - Prob. 10E
Ch. 7.3 - Prob. 1ECh. 7.3 - Prob. 2ECh. 7.3 - Prob. 3ECh. 7.3 - Prob. 4ECh. 7.3 - Prob. 5ECh. 7.3 - Prob. 6ECh. 7.3 - Prob. 7ECh. 7.3 - Prob. 8ECh. 7.4 - Prob. 1ECh. 7.4 - Prob. 2ECh. 7.4 - Prob. 3ECh. 7.4 - Prob. 4ECh. 7.4 - Prob. 5ECh. 7.4 - Prob. 6ECh. 7.4 - Prob. 7ECh. 7.4 - Prob. 8ECh. 7.4 - Prob. 9ECh. 7.4 - Prob. 10ECh. 7.4 - Prob. 11ECh. 7.4 - Prob. 12ECh. 7.4 - Prob. 13ECh. 7.6 - Prob. 1ECh. 7.6 - Prob. 2ECh. 7.6 - Prob. 3ECh. 7.6 - Prob. 4ECh. 7.6 - Prob. 5ECh. 7.6 - Prob. 6ECh. 7.6 - Prob. 7ECh. 7.6 - Prob. 8ECh. 7.6 - Prob. 9ECh. 7.6 - Prob. 10E
Knowledge Booster
Similar questions
- Which of the following molecules can have enantiomers? Identify any chiral carbon atoms. a. b. c.arrow_forward1. In the structure provided choose the option YES or NO to indicate if the carbon atoms are chiral. H 2. Assign the absolute configuration of the asymmetric/chiral centers in the provided structures a, b and c. a. a. C-H HO-CH3 H b. NH₂ 3. For the following two structures compared in parts A and B, determine whether they are enantiomers OR diastereomers OR the same structure drawn differently (non-bonding electrons not included for clarity). d. Br Br OH door OH Br b. CH3 HC OH H-C O Br $R= C. OH Br Br OH Br 1arrow_forwardOrganic Chemistry HW: 2,6-dimethyloct-2-ene cannot be handwritten, please type or use a program to draw Chiral Carbons show the expanded structure of this molecule. Determine if this molecule contains any chiral carbons. If there are chiral carbons in the molecule, circle or highlight all of them. If your molecule does not contain any chiral carbons explain why none of the carbons are chiralarrow_forward
- 7 For the following compound with two chiral centers assign 'R' and 'S' configurations. Draw enantiomers and diastereomers for the following compound? How many enantiomers and diastereomers are possible. Assign (R) and (S) configurations for all the structures. What are diastereomers? COOH H CI € Br -H CH3arrow_forwardHow many asymmetric/chiral centers are present in the molecule below?arrow_forwardDraw the optical isomers of CH3-CHOH-CHOH-CHO. Indicate the absolute configuration of each chiral center. From the drawn isomers, determine which are enantiomers and which are diastereomersarrow_forward
- Molecules without a plane of symmetry are chiral. In the model you constructed in no.1, the tetrahedral carbon is the stereocenter; the molecule is chiral. A simple test for a stereocenter in a molecule is to look for a stereocenter with four different atoms or groups attached to it; this molecule will have no plane of symmetry. label the stereocenter in each structure with an asterisk (*).arrow_forward7. Draw eight thiol isomers of the formula C3H12S. Identify chiral centers if any?arrow_forwardH ÇI CH3-C-COOH он CH3-C-CH2Br 1. ČI a b 81. Consider the structures above. Which of these two contains a stereocenter? Draw thestructure on your answer sheet and mark the stereocenter with an asterisk. 82. Which of these two is a chiral molecule? Explain your answer. 83. Which of these two can exist as a pair of enantiomers? 84. Draw the pair of enantiomers using three-dimensional representation.arrow_forward
- How to know if an atom is in sp3 when identifying chiral centers ?arrow_forwardBuild a model (attach a photo) for any of the following compounds. What is their relationship: (i) Identical in the same conformation, (ii) conformers, (iii) enantiomers, (iv) diastereomers, (v) constitutional (structural) isomers or none of (i) – (v)? H OH H. HO $Bu CI $Bu Relationship:arrow_forwardOptical Isomers Optical isomers have a chiral carbon atom. A chiral (or asymmetric) carbon atom is one that has four different groups attached to it. a H This carbon is chiral because it has four different groups attached to it. H,N CH, OH C - Identify which carbon (number) has the chiral center CI OH CH3 H H. H2N H- C-H H C F HICI Harrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co