
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
6th Edition
ISBN: 9781305687875
Author: Gilbert
Publisher: CENGAGE LEARNING - CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Question
Chapter 7.3, Problem 6E
Interpretation Introduction
Interpretation:
From the given two structures, the one which is
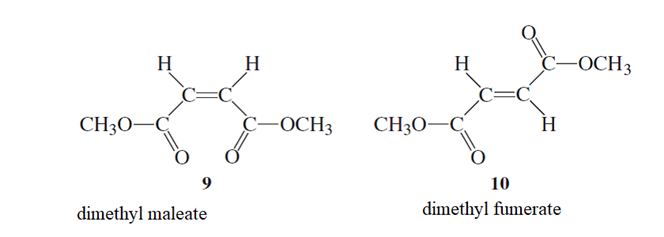
Cis − form trans − form
Concept Introduction :
The stereoisomers having different configurations at all stereocenters are known as enantiomers of each other and if there is a different configuration at only one stereocenter but same at other and both isomers are a non-mirror image of each other, they will be diastereomers.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Calculate the index of hydrogen deficiency of cyclohexene, C6H10, and account for this deficiency by reference to its structural formula.
(a) Give the (Z), (E), (R) or (S) configurations of each of the following alkenes.
Structure
Name
(i)
H
H,CH,C
CH3
(ii)
Cl
CH;Br
H;CH,CH,C
H,CH,CH,C
`H
(i) | Н,с,
CH,CH,OH
H
C(CH3)3
シII)
IIIC) trans-cyclooctene can be synthesized from cis-cyclooctene with UV light (hv).I
Use orbital line diagram to show electronic excitation and arrow-pushing to show
how this reaction can occur photochemically.
hy
Chapter 7 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
Ch. 7.2 - Prob. 1ECh. 7.2 - Prob. 2ECh. 7.2 - Prob. 3ECh. 7.2 - Prob. 4ECh. 7.2 - Prob. 5ECh. 7.2 - Prob. 6ECh. 7.2 - Prob. 7ECh. 7.2 - Prob. 8ECh. 7.2 - Prob. 9ECh. 7.2 - Prob. 10E
Ch. 7.3 - Prob. 1ECh. 7.3 - Prob. 2ECh. 7.3 - Prob. 3ECh. 7.3 - Prob. 4ECh. 7.3 - Prob. 5ECh. 7.3 - Prob. 6ECh. 7.3 - Prob. 7ECh. 7.3 - Prob. 8ECh. 7.4 - Prob. 1ECh. 7.4 - Prob. 2ECh. 7.4 - Prob. 3ECh. 7.4 - Prob. 4ECh. 7.4 - Prob. 5ECh. 7.4 - Prob. 6ECh. 7.4 - Prob. 7ECh. 7.4 - Prob. 8ECh. 7.4 - Prob. 9ECh. 7.4 - Prob. 10ECh. 7.4 - Prob. 11ECh. 7.4 - Prob. 12ECh. 7.4 - Prob. 13ECh. 7.6 - Prob. 1ECh. 7.6 - Prob. 2ECh. 7.6 - Prob. 3ECh. 7.6 - Prob. 4ECh. 7.6 - Prob. 5ECh. 7.6 - Prob. 6ECh. 7.6 - Prob. 7ECh. 7.6 - Prob. 8ECh. 7.6 - Prob. 9ECh. 7.6 - Prob. 10E
Knowledge Booster
Similar questions
- From the data in Figure 4-12 and Table 4-1, estimate the percentages of molecules that have their substituents in an axial orientation for the following compounds: (a) Isopropylcyclohexane (b) Fluorocyclohexane (c) Cyclohexanecarbonitrile, C6H11CNarrow_forwardEquilibrium Constants for Several Monosubstituted Cyclohexanes at 25 °C [equatorial] [axial] Keq Substituent Using the data in the table, calculate the percentage of molecules of chlorocyclohexane that have the Cl group in an equatorial position at 25 °C. H 1 CH3 18 Express your answer using two significant figures. CH3 CH2 21 (CH3 )2 CH % of equatorial conformer = % 35 (CH3 )3 C 4800 CN 1.4 F 1.5 Cl 2.4 Next > Br 2.2 I 2.2 НО 5.4arrow_forwardPredict the products for the hydrohalogenation reaction of 1-methyl-1,3-cyclohexadiene with 1 equivalent of HBr in thermodynamic conditions.arrow_forward
- The compound below is treated with chlorine in the presence of light. CH3 CH3CHCH,CH3 Draw the structure for the organic radical species produced by reaction of the compound with a chlorine atom. Assume reaction occurs at the weakest C-H bond. • You do not have to consider stereochemistry. You do not have to explicitly draw H atoms.arrow_forwardExamine each of the following ALLOWED electrocyclic, sigmatropic or cycloaddition reactions. Determine in each of these how many electrons are involved. For electrocyclic and cycloaddition reaction, this includes π-electrons (4n or 4n+2); for sigmatropic reactions, one σ bond electrons also contribures. Tell what type of concerted reaction each is, and whether 4n or 4n+2 system. Thank you!arrow_forwardThe compound below is treated with chlorine in the presence of light. CH3 CH,CHCH3 Draw the structure for the organic radical species produced by reaction of the compound with a chlorine atom. Assume reaction occurs at the weakest C-H bond. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms.arrow_forward
- Why does the first reaction, but not the second, give ring expansion?arrow_forwardCalculate the index of hydrogen deficiency for 1-hexene, C6H12, and account for this deficiency by reference to its structural formula.arrow_forwardWhat is the unsaturation number for compounds with the formula C6H10N3Br? O 2 O 3 O 4 O 1arrow_forward
- Using the table of average bond dissociation enthalpies at 25°C, determine which of the following reactions are energetically favorable at room temperature. Assume that ▲S = 0. Q. CH2=CH2 + CHI3 --> CH3CH2CI3arrow_forward3. According to the Hammond Postulate, what does transition state 3 (TS3) most closely resemble? First choice: Second choice: AG (a) TS2 (b) P (c) 12 (d) 11 TS2 TS1 M 11 Reaction Coordinate 12 TS3arrow_forward11. Compound X, (CH3)2CHCN, can be formed from a haloalkane, C3H7Br.(i) Name compound X............................................................................................................................(ii) Give the reagent and conditions necessary to form X from C3H7Br.Reagent..............................................................................................................Conditions..........................................................................................................(iii) Name and outline the mechanism for this reaction, showing clearly the structure of C3H7Br.Name of mechanism..........................................................................................Mechanism.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
