
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
6th Edition
ISBN: 9781305687875
Author: Gilbert
Publisher: CENGAGE LEARNING - CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Question
Chapter 7.4, Problem 12E
Interpretation Introduction
Interpretation: The GLC analysis of the oil needs to be explained which supports that there is a significant amount of a carbonyl-containing contaminant of carvone in the dill seed oil. It needs to be explained that how this proposal can be supported experimentally.
Concept Introduction: Carvone is known to be a natural product belonging to the class of terpenoids. It contains a
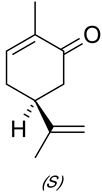
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
10.
(a) Remember the structures of cis- and trans-1,2-dibenzoylethylene. Which diastereomer
would have a greater affinity for the polar mobile solvent (phase) in a column chromatographic
separation?
(b) 5-6-7 refers to which solvent and name the compounds in that solvent.
(c) Define eluant.
Mescaline (see structure X) is a naturally-occuring psychedelic compound isolated from the peyote cactus. If
mescaline (X) was partitioned between 3M NaOH and dichloromethane (CH2CI2), would X partition primiarily into
the organic layer, aqueous layer, or neither layer?
H3CO.
„NH2
H3CO
mescaline
ÓCH3
Organic layer
Aqueous layer
O Neither layer. X would not partition effectively into either the aqueous or organic layer
Determine the % optical purity for phenyl succinic acid.
The weight of PSA is 0.25 g and the observed rotation is +3.90⁰.
The enantiomer is dissolved in 10 mL acetone and the optical rotation is taken in 1dm
Cell (length). You will only get credit if you show how, you obtained your results.
observed
'[a] max=+ 173.3 (c= 1, acetone) from Handbook. [a]-optical rotation / (length x conc)
% OP = ([a]/[a] max) x 100
Chapter 7 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
Ch. 7.2 - Prob. 1ECh. 7.2 - Prob. 2ECh. 7.2 - Prob. 3ECh. 7.2 - Prob. 4ECh. 7.2 - Prob. 5ECh. 7.2 - Prob. 6ECh. 7.2 - Prob. 7ECh. 7.2 - Prob. 8ECh. 7.2 - Prob. 9ECh. 7.2 - Prob. 10E
Ch. 7.3 - Prob. 1ECh. 7.3 - Prob. 2ECh. 7.3 - Prob. 3ECh. 7.3 - Prob. 4ECh. 7.3 - Prob. 5ECh. 7.3 - Prob. 6ECh. 7.3 - Prob. 7ECh. 7.3 - Prob. 8ECh. 7.4 - Prob. 1ECh. 7.4 - Prob. 2ECh. 7.4 - Prob. 3ECh. 7.4 - Prob. 4ECh. 7.4 - Prob. 5ECh. 7.4 - Prob. 6ECh. 7.4 - Prob. 7ECh. 7.4 - Prob. 8ECh. 7.4 - Prob. 9ECh. 7.4 - Prob. 10ECh. 7.4 - Prob. 11ECh. 7.4 - Prob. 12ECh. 7.4 - Prob. 13ECh. 7.6 - Prob. 1ECh. 7.6 - Prob. 2ECh. 7.6 - Prob. 3ECh. 7.6 - Prob. 4ECh. 7.6 - Prob. 5ECh. 7.6 - Prob. 6ECh. 7.6 - Prob. 7ECh. 7.6 - Prob. 8ECh. 7.6 - Prob. 9ECh. 7.6 - Prob. 10E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Acrolein (CH2=CH-CHO) shows two bands , at 210 nm (11,500) and 315 nm (15) . Assign these bands.arrow_forwardBesides polarimetry, name another method that could be used to assess the enantiomeric excess of a mixture of enantiomers. Be specific regarding the type of stationary phase or additive needed for your method to work.arrow_forwardAcetophenone is soluble in diethyl ether, isopropanol, and n-hexane. Identify and justify the most suitable solvent for UV-Vis characterizationarrow_forward
- Cis-3-hexene and trans-3-hexene can be separated using GC. Explain why these two compounds can be separated by GC and not by fractional distillation.arrow_forwardSagararrow_forwardGive a single benefit for each polymerisation from carrying them out under a nitrogen atmosphere? (d) Of the two polymers produced (A and B) one had Mn = 12,000 and a polydispersity of 1.1 and one a Mn = 11, 200 and a polydispersity of 1.7. Identify which set of molecular weight parameters corresponds to which polystyrene sample.arrow_forward
- Give and observation and inference when the compound hexane is subjected into test for Br2 in CCl4arrow_forwardAcrolein shows two bands at 210nm (11,500) and 315 nm(15).assign these bands.arrow_forwardConsider the chlorination (Cl2, hν) of optically active (2R,3R)-2-chloro-3-methylpentane. If the dichloro fraction is analyzed by gas liquid phase chromatography (GLPC), how many peaks may be seen? How many of the compounds in the dichloro fraction show optical activity?arrow_forward
- a) Indicate which absorption bands in the IR spectrum of the recrystallized product (meso-stilbene dibromide) are due to (i) Csp2 -H stretches, (ii) Csp3 -H stretches, and (iii) C=C stretches. b) The 1H NMR spectra in CDCl3 of dl-stilbene dibromide (Fig 1, 5.0-8.0 ppm region) and thepurified product, meso-stilbene dibromide (Fig 2) are shown below. Assign the peaks in thesespectra. c) Based on the NMR spectrum and melting point of your purified sample, derive an estimate ofthe purity (in mole %) of the material you have synthesized. Reaction: Trans-Stilbene + Bromine → Meso-Stilbene Dibromidearrow_forwardFor each of the topics below, please write a short paragraph of what it is and what it is used for: a) TLC and melting point b) IR spectroscopy c) UV-Vis spectroscopy d) NMR spectroscopyarrow_forwardPlease help with the following Organic Chemistry question regarding NMR for the compoundarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
Thermogravimetric Analysis [ TGA ] # Thermal Analysis # Analytical Chemistry Part-11# CSIR NET/GATE; Author: Priyanka Jain;https://www.youtube.com/watch?v=p1K-Jpzylso;License: Standard YouTube License, CC-BY