
(a)
Interpretation:
The bond which can be easily broken for the conversion of compound 6 to 7 and 8 needs to be determined.
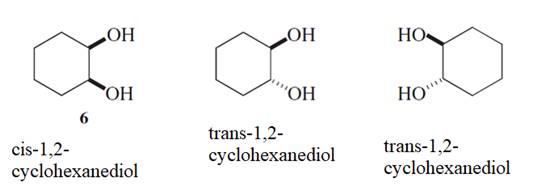
Concept Introduction :
Stereoisomers are the isomers with different spatial arrangement of the atoms, instead of order of atomic connectivity. It has the same molecular formula and the similar connectivity except for the procedure within 2D or 3D space. Like cis- and trans-but-2-ene both have two CH3 groups 2-H and a C=C but connectivity is different in the space.
(b)
Interpretation:
The value of
Concept Introduction :
Stereoisomers are the isomers with different spatial arrangement of the atoms, instead of order of atomic connectivity. It has the same molecular formula and the similar connectivity except for the procedure within 2D or 3D space. Like cis- and trans-but-2-ene both have two CH3 groups 2-H and a C=C but connectivity is different in the space.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- A. Electron-donating and Electron-withdrawing effects in conjugated structures 1. Go to the website http://molview.org/ 2. Draw the following structure on MolView. For each structure, obtain the following information: a. Charge b. Bond dipoles c. Over-all dipole Structure 1: Structure 2: H H. F F. Structure 3: Structure 4: Structure 5: Structure 6: You have to obtain the output from MolView. You can either copy the 3D image with the information, or you may draw them yourselves. Questions for Part A: 1. What are your observations based on the data you have obtained? 2. What can you infer about the nature of the substituents? Provide at least two inferences and explain your basis. 3. Which substituents is/are electron donating? Which of them are electron-withdrawing? 4. Which of the substituents is activating? Which ones are deactivating? Why do you say so? I0-arrow_forwardFind information on: 1. DDT 2. EDTA 3. orotic acid 4. сarboplatin 2. Propose the approximate shift/splitting pattern/etc. for each type of hydrogen that would be seen in an 'H-NMR spectrum Identify the # of each type of each carbon (i.e. how many peaks in the 13C spectra) 3. Identify infrared (IR) active bonds within each molecule 4.arrow_forward1. An aliphatic ketone absorbs at 1,715 cm-1. What is the frequency of this vibration in hertz, which is cycles per second or just per second, reciprocal seconds? 2. What is the energy equivalent of this stretching vibration in kcal/mole? 3. Why does 3,4-diethyl-3-hexene not have a carbon to carbon double bond stretching absorption band? 4. Why does a carbon to oxygen double bond absorption band have a greater intensity than a carbon to carbon double bond absorption band? 5. Using only IR, explain in detail how one could most easily differentiate between oct-1-ene and oct-1-yne if all carbon to carbon bonds are ignored. 6. Using only IR, explain in detail how one could most easily differentiate between butan-1-ol and butanoic acid.arrow_forward
- Which compound would you expect to to give rise to the mass spectra shown below? A. 1-Chloro-2-fluoroethene B. 2-chloroethanol C. buthylchloride D. Isopropyl chloride E. isopropyl bromidearrow_forwardb. Calculate the max of the given compounds with the help of Woodward's Fiesher rule.arrow_forward1. a. Why would a column be ineffective if you chose an eluting solvent that was too polar? b. Why would a column be ineffective if you chose an eluting solvent that was not polar enough?arrow_forward
- 4. Place the following bonds in order of increasing wave number as observed in their infrared spectrum for the following functional groups and explain why you chose this order. A) Alkane, alkene, alkyne (ignore any bonds to hydrogen) B) O-H and 0-D bonds (D = Deuterium) (25)arrow_forwardProvide the following from NBS and under UV light: N-Br hv. CCI, D. B. Br A. Br Br Br O a. choice A. O b. choice B. Oc. choice C. O d. choice D.arrow_forwardB. How can you tell the difference between an aromatic ring double bond and an alkene double bond on an ir spectrum?arrow_forward
- 6. IR spectroscopy experiment is used to evaluate the success of the reaction. The IR spectra for acetaminophen and phenacetin are shown below. a. Identify which spectrum is for acetaminophen and which is for phenacetin. b. Label the key bond/functional group signals on each spectrum. (eg N-H, C=0, O-H, C=C, C(sp2)-H, C(sp3)-H) OH N H acetaminophen phenacetin 100.0 90 80 70 60 30 40 30 20 10 0.0 40000 3000 2000 1500 1000 400.0 em-1 LOD D 4D00 3000 s0o 2000 1500 1000arrow_forwardBelow, t-butyl chloride (molecular ion = 92) has been marked for "splitting" in MS, just as in the lecture. Find the correct values for A, B, C, and D. NOTE: B and D have more than one correct value because of something in the molecule!arrow_forward2. Kindly give what is asked in these problems.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





