
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
6th Edition
ISBN: 9781305687875
Author: Gilbert
Publisher: CENGAGE LEARNING - CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Question
Chapter 7.2, Problem 4E
Interpretation Introduction
Interpretation:
The meso compounds needs to be determined from the given set of compounds.
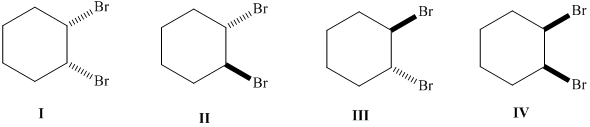
Concept Introduction :
A meso compound is defined as a compound having two or more stereocenters along with an internal plane of symmetry.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please show all arrow pushing mechaarsas.
Thank you!
1. What are the similarities and
differences of candles and
plastic milk containers?
2.Why does the Tm increase with
more C atoms in chain?
10) Can H3PO4 exist with H₂PO4 in a mixture?
What about H₂PO4 and HPO4²?
What about H₂PO4 and PO,³?
Chapter 7 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
Ch. 7.2 - Prob. 1ECh. 7.2 - Prob. 2ECh. 7.2 - Prob. 3ECh. 7.2 - Prob. 4ECh. 7.2 - Prob. 5ECh. 7.2 - Prob. 6ECh. 7.2 - Prob. 7ECh. 7.2 - Prob. 8ECh. 7.2 - Prob. 9ECh. 7.2 - Prob. 10E
Ch. 7.3 - Prob. 1ECh. 7.3 - Prob. 2ECh. 7.3 - Prob. 3ECh. 7.3 - Prob. 4ECh. 7.3 - Prob. 5ECh. 7.3 - Prob. 6ECh. 7.3 - Prob. 7ECh. 7.3 - Prob. 8ECh. 7.4 - Prob. 1ECh. 7.4 - Prob. 2ECh. 7.4 - Prob. 3ECh. 7.4 - Prob. 4ECh. 7.4 - Prob. 5ECh. 7.4 - Prob. 6ECh. 7.4 - Prob. 7ECh. 7.4 - Prob. 8ECh. 7.4 - Prob. 9ECh. 7.4 - Prob. 10ECh. 7.4 - Prob. 11ECh. 7.4 - Prob. 12ECh. 7.4 - Prob. 13ECh. 7.6 - Prob. 1ECh. 7.6 - Prob. 2ECh. 7.6 - Prob. 3ECh. 7.6 - Prob. 4ECh. 7.6 - Prob. 5ECh. 7.6 - Prob. 6ECh. 7.6 - Prob. 7ECh. 7.6 - Prob. 8ECh. 7.6 - Prob. 9ECh. 7.6 - Prob. 10E
Knowledge Booster
Similar questions
- How many stereocenters are in the D-glucose molecule? a b. 2 . C 0 d 1 Clear my choice ROOM How many Rand S stereocenters are there in the 5-a Dihydrotestosterone molecule? a. 45, 38 b. 15, 6R C35, 4R d. 35, 3R e. 65 1R Clear my choice Total number of stereoisomers possible for the following compounds s O FORMIGA 0 270arrow_forward7. Why are protecting groups needed in this reaction and what is the purpose of CH2C12? Can you explain this reaction to me? I understand intermediate 10 will be a disaccharide. Bno Bno o co, Bno OAc NH TMSOTI (1,2)-linked tetramannoside 10 CH,Clz Bno. он Bno o Bno ROAIarrow_forward12. Using a suitable example, explain (using structures) what is meant by a meso compound. Draw the mirror image of your meso compound. Can a meso compound have an enantiomer? Explain the reason for your answer||arrow_forwardThe structure given below is a tetrasaccharide. i) For each pair of sugars (I-II, Il-III, III-IV), identify the type of linkage (i.e., 1→4, 1→6, etc.) that connects them. You do not have to specify whether the linkage is alpha or beta. ii) Identify all sugars that are in a furanose form. ОН ОН I HO- OH ОН О II Но OH Но НО HO- II ОН ОН IVarrow_forward2. If curve 3 in the figure to the right represents the binding behavior of normal Hb in the presence of 2 mM 2,3-Bisphophoglycerate (BPG), which curve represents the binding behavior of Hb at 4 mM BPG? A) 4 B) 3 C) 2 D) 1 E) None of the above 3. How many stereoisomers are possible for N-acetyl glucosaminopyranose? A) 4 B) 8 C) 16 D) 24 E) 32 % saturation 2 3 4 pp oxygen (Tom)arrow_forwardFind an example of a chiral molecule in biology whose enantiomers have markedly different functions. Where is the chiral center located, and how can it be identified? Find and include an image of its molecular structure. What function does the active enantiomer play biologically, and how is this function hindered or changed with the other enantiomer? Are there any synthetic considerations for this molecule?arrow_forwardWhich of the molecules shown are meso/ mesomeric? b. NH HN NH HN HO d. SCH, HO,C SCH, All four molecules are meso. Molecules b and d. Molecules a, b, and d. Molecules c and d. Molecules a and d.arrow_forward3 Which is the stereochemical relationship between this pair of molecules?arrow_forwardExamine the structure below and (a) draw all its stereoisomers, as well as (b) indicate the relationship between all the stereoisomers as either (i) enantiomers, (ii) diastereomers, or (iii) meso compounds. Br OHIarrow_forwarda) Which structures are Enantiomers? b) Which structures are Diastereomers? c) Which structures are meso compounds? Use the letters only for your answers. А. В. H H CI H H Cl C. H H D. H Cl ČI Cl CI Harrow_forwardA. Construct two models of 1-bromo-1-chloroethane. Test to see if the two models are superimposable on one another. If they are superimposable, change the position of Br and Cl on one model. Now you have two models that are not superimposable. These two are enantiomers, non-superimposable mirror image isomers. (Turn, twist, rotate, do anything that you want to these models except interchange the positions of the groups; they remain non-superimposable.) Position these enantiomers so that they appear as mirror images. Arrange them so that the Br is on top and the CH3 is on the bottom of the chiral carbon atom, and the H and Cl are coming out of the plane toward you. Now draw the projection formulas for these enantiomers. Brarrow_forward-Br How many chiral centers are present in this molecule. Identify the chiral center. Mark with x. How many stereoisomers are possible.arrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY