
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
6th Edition
ISBN: 9781305687875
Author: Gilbert
Publisher: CENGAGE LEARNING - CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Question
Chapter 7.3, Problem 1E
Interpretation Introduction
Interpretation:
The exclusion of thermal charge of pi bond needs to be determined as the starting step for the isomerization of 9 to 10.
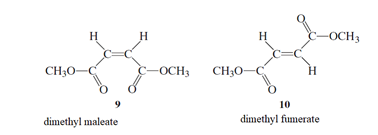
Concept Introduction :
Stereoisomers are the isomers with different spatial arrangement of the atoms, instead of order of atomic connectivity. It has the same molecular formula and the similar connectivity except for the procedure within 2D or 3D space. Like cis- and trans-but-2-ene both have two CH3 groups 2-H and a C=C but connectivity is different in the space.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Do both plz dont give incomplete solution
DO BOTH MUST
Help ASAP
Illustrate the resonance effect of the methoxy group -OCH3, on the structure of the benzene ring. Do this by writing all the possible resonance forms for methoxybenzene, including the hybrid.
Based on your structures, explain how the presence of the -OCH3 group affects: (i) the reactivity of the benzene ring towards electrophilic attack; (ii) the orientation or point of attack of an incoming electrophilic reagent on the benzene ring.
How would I go about determining the mechanism of this problem?
Chapter 7 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
Ch. 7.2 - Prob. 1ECh. 7.2 - Prob. 2ECh. 7.2 - Prob. 3ECh. 7.2 - Prob. 4ECh. 7.2 - Prob. 5ECh. 7.2 - Prob. 6ECh. 7.2 - Prob. 7ECh. 7.2 - Prob. 8ECh. 7.2 - Prob. 9ECh. 7.2 - Prob. 10E
Ch. 7.3 - Prob. 1ECh. 7.3 - Prob. 2ECh. 7.3 - Prob. 3ECh. 7.3 - Prob. 4ECh. 7.3 - Prob. 5ECh. 7.3 - Prob. 6ECh. 7.3 - Prob. 7ECh. 7.3 - Prob. 8ECh. 7.4 - Prob. 1ECh. 7.4 - Prob. 2ECh. 7.4 - Prob. 3ECh. 7.4 - Prob. 4ECh. 7.4 - Prob. 5ECh. 7.4 - Prob. 6ECh. 7.4 - Prob. 7ECh. 7.4 - Prob. 8ECh. 7.4 - Prob. 9ECh. 7.4 - Prob. 10ECh. 7.4 - Prob. 11ECh. 7.4 - Prob. 12ECh. 7.4 - Prob. 13ECh. 7.6 - Prob. 1ECh. 7.6 - Prob. 2ECh. 7.6 - Prob. 3ECh. 7.6 - Prob. 4ECh. 7.6 - Prob. 5ECh. 7.6 - Prob. 6ECh. 7.6 - Prob. 7ECh. 7.6 - Prob. 8ECh. 7.6 - Prob. 9ECh. 7.6 - Prob. 10E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the species below best depicts the likely transition state for the reaction between methyl iodide, CH3I and sodium methylthiolate, NaSCH3?arrow_forwardPlease don't provide handwritten solution ......arrow_forwardSuppose the compound is synthesized via EAS reaction of diazonium salt and coupling agent. Determine the ff. 1. Structure of diazonium salt and coupling agent 2. Mechanism of the coupling reaction НО. N= Narrow_forward
- Which one of the following statements is FALSE? O The formation of a Grignard reagent requires use of 1 molecule of magnesium metal for every 1 molecule of alkyl bromide used. Organometallic compounds undergo transmetallation to form a less polar carbon-metal bond. O Reductive elimination is the last step in a Pd-mediated cross-coupling reaction. Oxidative addition is when a metal inserts between a carbon-halogen bond. The formation of an organolithium reagent requires use of 1 molecule of lithium metal for every 1 molecule of alkyl bromide used.arrow_forwardPropose a reaction mechanism for P4S3 + I2 isomerizationarrow_forwardEXPLAIN BY WORDS IN ORDER FOR ME TO UNDERSTAND THE GENERAL REACTION SCHEME. IN THIS CROSS-COUPLING REACTION WHICH ORGANOHALIDE (R-X) REACTS WITH ORGANOBORANE [R1B(OR)2].arrow_forward
- Please give a reasonable mechanism for the following reaction using a multiple component reactions. Please draw it out and write out the steps. PLEASE SHOW WITH ARROWS. 5 PhNH + n-Bu 10 mol% TI (NM)2(doma) + ABU-NC tol 100 °C, 48 h, 77% Bu n-Buarrow_forwardwrite the mechanism of this reactionarrow_forwardCan you do 5,6,7 and explain it? State the mechanism in which through reaction proceeds sn2 or sn1 and can you explain the process of the mechanismarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
SAR of Anticancer(Antineoplastic) Drug/ Alkylating agents/ Nitrogen Mustard; Author: Pharmacy Lectures;https://www.youtube.com/watch?v=zrzyK3LhUXs;License: Standard YouTube License, CC-BY