
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
6th Edition
ISBN: 9781305687875
Author: Gilbert
Publisher: CENGAGE LEARNING - CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 7.3, Problem 2E
Interpretation Introduction
Interpretation:
The reason for the exclusion of direct photo excitation of 9 in order to convert to 10 needs to be explained.
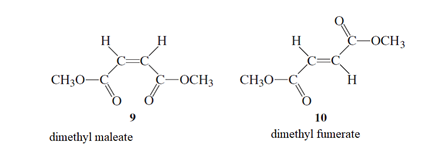
Cis − form trans - form
Concept Introduction :
Stereoisomers are the isomers with different spatial arrangement of the atoms, instead of order of atomic connectivity. It has the same molecular formula and the similar connectivity except for the procedure within 2D or 3D space. Like cis- and trans-but-2-ene both have two CH3 groups 2-H and a C=C but connectivity is different in the space.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What structural features of Hydroxypropyl Acrylate (HPA) allow it to be detected by HPLC?
Absorption bands in the region of 1600-1700 cm-1 are often used for quantification of caffeine. What does the bands in this region refer to?
Why do aldehydes, esters, and amides all have a strong absorption in the
1630-1780 cm1 region of their IR spectra?
A) The bond between H and the sp³-hybridized C in these functional groups
vibrates in this energy range.
B) Each of these functional groups has at least two resonance structures, and
the different vibrations of the resonance structures give off energy in this
region.
C) The bond between O and the sp²-hybridized C in these functional groups
vibrates at a frequency in this energy range.
D) Light at this wavenumber causes the average C to O bond length to increase
which causes more of this light to be transmitted.
E) An electron in the bond of these functional groups gets excited to the *
orbital.
Chapter 7 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
Ch. 7.2 - Prob. 1ECh. 7.2 - Prob. 2ECh. 7.2 - Prob. 3ECh. 7.2 - Prob. 4ECh. 7.2 - Prob. 5ECh. 7.2 - Prob. 6ECh. 7.2 - Prob. 7ECh. 7.2 - Prob. 8ECh. 7.2 - Prob. 9ECh. 7.2 - Prob. 10E
Ch. 7.3 - Prob. 1ECh. 7.3 - Prob. 2ECh. 7.3 - Prob. 3ECh. 7.3 - Prob. 4ECh. 7.3 - Prob. 5ECh. 7.3 - Prob. 6ECh. 7.3 - Prob. 7ECh. 7.3 - Prob. 8ECh. 7.4 - Prob. 1ECh. 7.4 - Prob. 2ECh. 7.4 - Prob. 3ECh. 7.4 - Prob. 4ECh. 7.4 - Prob. 5ECh. 7.4 - Prob. 6ECh. 7.4 - Prob. 7ECh. 7.4 - Prob. 8ECh. 7.4 - Prob. 9ECh. 7.4 - Prob. 10ECh. 7.4 - Prob. 11ECh. 7.4 - Prob. 12ECh. 7.4 - Prob. 13ECh. 7.6 - Prob. 1ECh. 7.6 - Prob. 2ECh. 7.6 - Prob. 3ECh. 7.6 - Prob. 4ECh. 7.6 - Prob. 5ECh. 7.6 - Prob. 6ECh. 7.6 - Prob. 7ECh. 7.6 - Prob. 8ECh. 7.6 - Prob. 9ECh. 7.6 - Prob. 10E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The molecular ion region of the spectrum of biphenyl. M1 is observed at m/z 154 and the intensity of M 1 1 is 12.9% of M1. What formulas of the type CnHxOyNz are consistent with the spectrum?arrow_forwardWhy C=O in concentrated/pure aliphatic carboxylic acid appear at lower stretching frequency (1710 cm-1) than dilute solution (1760 cm-1)?arrow_forward1.) The "m/z value" and the" relative abundance " for M+2 peak in (CH3)2CH-Br?arrow_forward
- What can be said about the ratios of the multiplet areas in the 13C spectra for trichloroethene; 1,2-dichloroethane; 1,1,1-trichloroethane; 1,1,2-trichloroethane.arrow_forward[References] The mass spectrum of octanoic acid, CH3(CH2)6COOH, exhibits a series of peaks differing by 14 amu at m/z 29, 43, 57, 71, 85, and 99. Draw the structure of the fragment resulting in the m/z 71 peak. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. • Do not use the square brackets tool in your answer. opy aste H,C earch 84%arrow_forwardFollowing is the mass spectrum of 1-bromobutane. Q.) Account for the appearance of the M + 2 peak of approximately 95% of the intensity of the molecular ion peak.arrow_forward
- Explain why a ketone carbonyl typically absorbs at a lower wavenumber than an aldehyde carbonyl (1715 vs. 1730 cm−1).arrow_forwardFill in the table by matching the numbers of the structure to their corresponding peaks of the C-13 spectra.arrow_forwardThe following Infra-Red Spectrum is that of an unknown organic compound. Infrared Spectrum 1.0 0.8 0.6 0.4 0.2 0.0 3500 3000 2500 2000 1500 1000 500 Wavenumbers (cm-1) The compound could be: butan-2-one а. b. methyl ethanoate c. propanoic acid d. ethanol Transmitancearrow_forward
- Account for the presence of the following peak in the mass spectrum of hexanoic acid, CH3(CH2)4COOH. Q.) A series of peaks differing by 14 amu at m/z 45, 59, 73, and 87arrow_forwardDraw the instrumentation of ATR spectroscopy? Please answer at your own words.arrow_forwardно Chemical Proton Integration Splitting Shift (8)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY