
Concept explainers
(a)
Interpretation:
Whether the given pair of 1, 2-dibromocyclohexane is enantiomers or diastereomer needs to be determined. This also needs to be indicated whether each pair haveidentical or different physical properties.
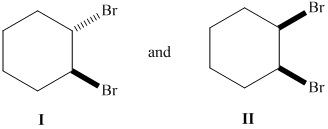
Concept Introduction :
The stereoisomers having different configurations at all stereocenters are known as enantiomers of each other and if there is a different configuration at only one stereocenter but same at other and both isomers are the non-mirror image of each other, they will be diastereomers.
(b)
Interpretation:
Whether the given pair of 1, 2-dibromocyclohexane is enantiomers or diastereomer needs to be determined. This also needs to be indicated whether each pair haveidentical or different physical properties.
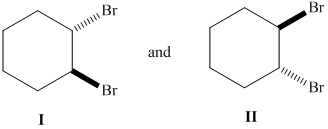
Concept Introduction :
The stereoisomers having different configurations at all stereocenters are known as enantiomers of each other and if there is a different configuration at only one stereocenter but same at other and both isomers are the non-mirror image of each other, they will be diastereomers.
(c)
Interpretation:
Whether the given pair of 1, 2-dibromocyclohexane is enantiomers or diastereomer needs to be determined. This also needs to be indicated whether each pair haveidentical or different physical properties.
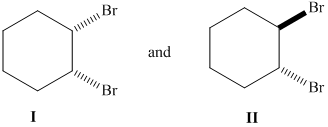
Concept Introduction :
The stereoisomers having different configurations at all stereocenters are known as enantiomers of each other and if there is a different configuration at only one stereocenter but same at other and both isomers are the non-mirror image of each other, they will be diastereomers.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- Ketones react with alcohols to yield products called acetals. Why does the all-cis isomer of 4-tert-butyl-1,3-cyclohexanediol react readily with acetone and an acid catalyst to form an acetal, but other stereoisomers do not react? In formulating your answer, draw the more stable chair conformations of all four stereoisomers and the product acetal for each one.arrow_forwardFollowing is a planar hexagon representation of L-fucose, a sugar component of the determinants of the A, B, O blood group typing. For more on this system of blood typing, see Chemical Connections: A, B, AB, and O Blood Group Substances in Chapter 25. (a) Draw the alternative chair conformations of L-fucose. (b) Which of them is more stable? Explain.arrow_forwardDraw Newman projections of 1,2-ethanediol for all eclipsed conformations formed by rotation of 0° to 360° about carbon-carbon single bond. Which eclipsed conformation(s) has (have) the lowest and highest energy?arrow_forward
- Consider 1-bromo-2-methylpropane and draw the following. Q) The staggered conformation(s) of highest energyarrow_forwardHow the relative stability of the two conformations of any disubstituted cyclohexane can be analyzed ?arrow_forwardThe following are representations of two forms of glucose. The six-membered ring is known to exist in a chair conformation in each form. Draw clear representations of the most stable conformation of each. Are they two different conformations of the same molecule, or are they stereoisomers that cannot be interconverted by rotation about single bonds? Which substituents (if any) occupy axial sites?arrow_forward
- Consider 1-bromo-2-methylpropane and draw the following. Q) The staggered conformation(s) of lowest energyarrow_forwardIdentify the stereochemistry of this molecule.arrow_forwardHow to determine stereochemistry of disubstitued cyclohexane molecules, and if they are meso, enantiomers, or diasteroemers?arrow_forward
- assign the stereochemical configuration of the selected tetrahedral carbon chiral centers (R, S or N (not a chiral center)) and the alkene (E, Z or N (not a stereocenter)) that are indicated by the arrows (note that you do not have to assign the configuration of every chiral center in the molecule). If the atom in question is not a chiral center or is not a stereocenter circle N for neither.arrow_forwardConsider 1-bromopropane, CH3CH2CH2Br. (a) Draw a Newman projection for the conformation in which CH3 and -Br are anti (dihedral angle 180°). (b) Draw Newman projections for the conformations in which - CH3 and -Br are gauche (dihedral angles 60° and 300°). (c) Which of these is the lowest energy conformation? (d) Which of these conformations, if any, are related by reflection?arrow_forward(a) Draw all stereoisomers formed by monochlorination of the cis and trans isomers of 1,2-dimethylcyclobutane drawn below. (b) How many constitutional isomers are formed in each reaction? (c) Label any pairs of enantiomers formed.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


