
(a)
Interpretation:
The reagent and condition is to be proposed for step 1.
Concept introduction:
Friedal – Crafts alkylation (or acylation):
Friedal – Crafts alkylation (or acylation) is one of the electrophilic substitution reaction.

(b)
Interpretation:
The reagent and condition is to be proposed for step 2 and step 3.
Concept introduction:
Oxime formation:
(c)
Interpretation:
The reductive amination is to be explained and the explanation has to be given for two step synthesis of amination is used rather than reductive amination.
Concept introduction:
Reductive amination reaction: Amination is the process by which an
The conversion of Carbonyl group in to amine via imine intermediate is called reductive amination.
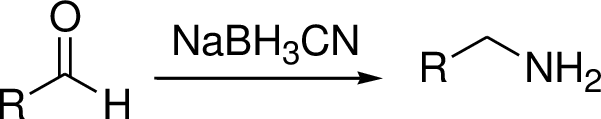
(D)
Interpretation:
The reagent and condition is to be proposed for step 4 and step 5.
Concept introduction:
Reduction: Aldehydes or ketones undergoing reduction by using reducing agent like LAH or NaBH4 which provides alcohol.
(E)
Interpretation:
The possible stereoisomer’s has to be shown if the product is chiral.
Concept introduction:
Isomer: A molecule having the same molecular formula but with different chemical structure is called isomer.
Enantiomers: A compound which is non-superimposable mirror image is called enantiomers.
Diastereomers: A compound which is non-superimposable and non-mirror image is called diastereomers.
Racemic mixture: A racemic mixture is simply a mixture containing an equal amount of each enantiomer.
Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
Trending nowThis is a popular solution!

Chapter 23 Solutions
Organic Chemistry
- Following are 1H-NMR spectra for compounds B (C6H12O2) and C (C6H10O). Upon warming in dilute acid, compound B is converted to compound C. Deduce the structural formulas for compounds B and C.arrow_forwardFollowing is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forward
- Phenacetin is an analgesic compound having molecular formula C10H13NO2. Once a common component in over-thecounter pain relievers such as APC (aspirin, phenacetin, caffeine), phenacetin is no longer used because of its liver toxicity. Deduce the structure of phenacetin from its 1H NMR and IR spectra.arrow_forward(b) State the reagents needed to convert benzoic acid into the following compounds. (i) C6H§COCI (ii) C,H$CH2OH (iii) C6H$CONHCH3arrow_forwardPropose single-step and multistep syntheses of acid derivatives from compoundscontaining other functional groups. Propose multistep syntheses using acid derivatives as starting materials and intermediates.arrow_forward
- (b) Propose a synthesis of (2,2-dimethylpropyl)benzene from benzene.arrow_forward(a) Explain how NaBH, in CH;OH can reduce hemiacetal A to 1,4-butanediol (HOCH,CH,CH,CH,OH). (b) What product is formed when A is treated with Ph;P=CHCH,CH(CH),? (c) The drug isotretinoin is formed by reaction of X and Y. What is the structure of isotretinoin? Although isotretinoin (trade name Accutane or Roaccutane) is used for the treatment of severe acne, it is dispensed under strict controls because it also causes birth defects. PPha NaOCH,CH3 HO- isotretinoin HO A Br X Yarrow_forwardThe following questions concern ethyl (2-oxocyclohexane)carboxylate.(a) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by a Dieckmann cyclization.(b) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by acylation of a ketone.(c) Write structural formulas for the two most stable enol forms of ethyl (2-oxocyclohexane)carboxylate.(d) Write the three most stable resonance contributors to the most stable enolate derived from ethyl (2-oxocyclohexane)carboxylate.(e) Show how you could use ethyl (2-oxocyclohexane)carboxylate to prepare 2-methylcyclohexanone.(f) Give the structure of the product formed on treatment of ethyl (2-oxocyclohexane)-carboxylate with acrolein (H2C=CHCH=O) in ethanol in the presence of sodium ethoxidearrow_forward
- (c)Show step by step how to synthesize methoxybenzene from benzene.arrow_forwardPropose a synthesis of the anti-inflammatory drug Ibuprofen from benzene. Show all reagents and all intermediate structures. Assume that ortho and para isomers can be separatedarrow_forwardProvide reagents/conditions to accomplish the following syntheses. More than one step is required in some cases.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

