
Concept explainers
a.
Interpretation:
For the below compound the chirality centers has to be labeledand number of chiral centers should be determined.
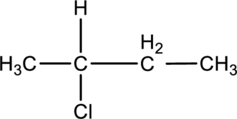
Concept introduction:
Chirality center:
When a carbon atom is bonded to four different groups, then that carbon is named as a chiral carbon and it is called as chirality center. Generally, the carbon atom contains tetrahedral valency, therefore it bonded to four atoms or molecules. The chiral center is bonded with four different elements or groups. If the single carbon atom is bonded to two or more identical group or atom, then it is also not considered as chiral carbon. In addition to this, if the carbon atom contains multiple bond then it does not able to bond with four different groups then itis not consideredto be as a chirality center.
b.
Interpretation:
For the below compound the chirality centers has to be labeled and number of chiral centers should be determined.
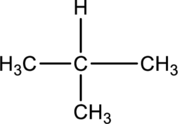
Concept introduction:
Refer to part “a”.
c.
Interpretation:
For the below compound the chirality centers has to be labeled and number of chiral centers should be determined.
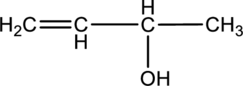
Concept introduction:
Refer to part “a.”.
d.
Interpretation:
For the below compound the chirality centers has to be labeled and number of chiral centers should be determined.
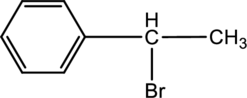
Concept introduction:
Refer to part ‘a.’.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Principles of General, Organic, Biological Chemistry
- Modify the existing drawings to draw both stereoisomers of this compound. H₂C₂ CH₂ HO OH H H₂C Edit Drawing CH3 HO OH Harrow_forwardA) ОН B) HO. Molecule A is achiral and molecule B is chiral Molecule A is chiral and molecule B is achiral Both molecules are achiral. Both molecules are chiral.arrow_forwardLocate the stereogenic centers in each molecule. Compounds may have one or more stereogenic centers.arrow_forward
- Label each compound as chiral or achiral. Compounds that contain a single carbon common to two rings are called spiro compounds. Because carbon is tetrahedral, the two rings are perpendicular to each other.arrow_forwardHow many chirality centers are in each compoundarrow_forward1a. How many stereogenic centers are present 1c. Draw a three-dimensional structure of a in the structure below? Indicate them with asterisk(s). How many stereoisomers stereoisomers are possible? chiral compound with the molecular formula of C4H4Cl₂ that does not have a stereogenic carbon. In addition, draw the enantiomer of this compound. Number of stereogenic centers: Number of stereoisomers possible: 1b. Draw one of the two most stable stereoisomers of the compound in 1a using a planar structure with wedges and dashes. Now draw it in its preferred chair conformation. 1d. Draw two meso compounds with the molecular formula of C7H14.arrow_forward
- Place an asterisk (*) next to the chirality center (sometimes called chiral atom) or centers in each compound. It is possible that a compound may not have a chiral center.arrow_forwardLabel the chirality center (if one exists) in each compound. A compound may contain zero or one chirality center.arrow_forwardLabel each compound as chiral or achiral.arrow_forward
- What is the relationship between the two molecules shown below? OH H CH3 ОН H H H H H Н. H. H. -CH3 H H H H H H A) enantiomers B) diastereomers C) conformational isomers D) constitutional isomers E) identicalarrow_forwardThere are eight chirality centers in the following molecule. Identify each asymmetric carbon atom by clicking on the circled carbons that are chiral. HOarrow_forwardC. Smelling Chirality. Carvone Olfactory receptors in our noses, used for the detection of odors, are chiral. Therefore, in some cases, we can tell the difference between enantiomers by their odor. Carvone is one such compound that we can distinguish between the R and S enantiomer by their odor. 1. Identify the chiral center in carvone and draw the R and S enantiomers in the indicated boxes. 2. Smell both of the enantiomers of carvone and provide a brief description of their odors in the same boxes. Carvone is perfectly safe to smell as both enantiomers are found in various foods. R-carvone S-carvonearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
