
a.
Interpretation:
The increasing order of the boiling points of the below compounds has to be determined.
Boiling point of
Alkyl group with one halogen atom is a polar molecule and has net dipole; therefore, it has higher boiling and melting points than normal
b.
Interpretation:
The increasing order of the boiling points of the below compounds has to be determined.
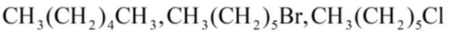
Concept introduction:
Refer to part a.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Principles of General, Organic, Biological Chemistry
- In each pair of compounds, pick the one with the higher boiling point. Explain your reasoning.a. CH3OH or CH3SHb. CH3OCH3 or CH3CH2OHc. CH4 or CH3CH3arrow_forwardArrange the compounds of each group in order of increasing boiling point. a. 1-butanol, 1-pentanol, 1-propanol b. 1,2-butanediol, 2-butanol, pentanearrow_forwarda. Which of the following compounds forms hydrogen bonds between its molecules? 1. CH3CH2OCH2CH2OH 2. CH3CH2N(CH3)2 3. CH3CH2CH2CH2Br 4. CH3CH2CH2NHCH3 5. CH3CH2CH2COOH 6. CH3CH2CH2CH2F b. Which of the preceding compounds forms hydrogen bonds with a solvent such as ethanol?arrow_forward
- Arrange the compounds in order of increasing boiling point. HO. B C D Aarrow_forwardH. E. G но но HO B. C HO ONa HO 2.52 Rank molecules A-H in order from lowest to highest boiling point. 2.8 Ranking Boiling Points and Solubilities of Structurally Similar Compoundsarrow_forward1. Which of the following has the highest boiling point? HCOOH CH3COOH CH3CH2COOH C6H5COOH 2. Which of these compounds would have the highest boiling point? CH3CH2OCH2CH2OCH3 CH3CH2OCH2OCH2CH2 CH3OCH2CHOCH3 HOCH2CH2CH2CH2CH2OH CH3OCH2CH2CH2OCH3arrow_forward
- a. Draw two different structures with the molecular formula C,H,0. b. Name the functional group in each structure. c. Which one will have the higher boiling point, and why?arrow_forwardFor each set of compounds, which compound has the highest boiling point?a. (i) Hexane, (ii) octane, or (iii) decaneb. (i) (CH3)3C-C(CH3)3 or (ii) (CH3)2CH-CH2CH2-CH(CH3)2arrow_forwardWhich one of the following ranks the compounds in order of lowest boiling point to highest boiling point based only on intermolecular forces? A.)CH3CH2CH3 < CH3OCH3< CH3CH2CH3 B.)CH3OCH3 < CH3CH2CH3 < CH3CH2OH C.)CH3CH2CH3 < CH3CH2CH3 <CH3OCH3 D.)CH3CH2CH3 < CH3OCH3 < CH3CH2OHarrow_forward
- ● Arrange these compounds in order of increasing boiling point. Heptane HO. LOH 1,5-Pentanediol Propanol 1-Hexanol LOHarrow_forward34. Which of these generic structures denotes an ether? G. RCR В. R-OH C. R-O-R' E. RCH F. A. R-CI D. R-NH-R' RCOHarrow_forwardRank the following by decreasing boiling points, so that 1 is the highest and 3 is the lowest. 1. CH3-O-CH3 2. CH3-CH2-OH 3. CH3-CH2-CH3arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





