
a.
Interpretation:
For a given compound type of alkyl halide has to be determined.
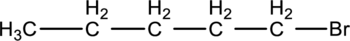
Classification of
The organic molecules contains halogen atoms is called alkyl halides. Generally, the halogen atom is denoted as
b.
Interpretation:
For a given compound type of alkyl halide has to be determined.
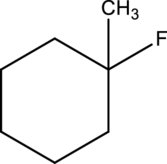
Concept introduction:
Refer to part a.
c.
Interpretation:
For a given compound type of alkyl halide has to be determined.
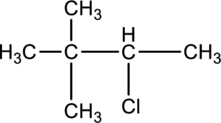
Concept introduction:
Refer to part a.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Principles of General, Organic, Biological Chemistry
- Which alkyl halide has the highest boiling point? A. CH3BrB. CH3FC. CH3ClD. CH3larrow_forward1. Name the following ether. 2. Name the following thiol. C C-C-C-C-0-C-C -9-9-⁹ C-C-C-C-C-S-H C-C-C-C 3. Will the ether H bond with water? Will the thiol H bond with water?arrow_forwardInstructions: Draw out each compound to clearly show what groups are bonded to the carbonyl carbon. Label each compound as a carboxylic acid, ester, or amide. a. CH3CH2CO2CH2CH3 b. CH3CONHCH3 c. (CH3)3CCO2H d. (CH3)2CHCON(CH3)2 Instructions: Give the IUPAC name for each compound. A. CH₂ CH₂CH₂CH₂CCH₂COOH CH3 B. CH₂CHCH₂CH₂COOH CH₂COOH CH₂CH3 C. (CH,CH,),CHCH,CHCOOH Instructions: Give the structure corresponding to each IUPAC name. a. 2-bromobutanoic acid b. 2,3-dimethylpentanoic acid c. 2-ethyl-5,5-dimethyloctanoic acid d. 3,4,5,6-tetraethyldecanoic acidarrow_forward
- Classify each alkyl halide as 1 °, 2 °, or 3 °.arrow_forwardClassify each OH group in dexamethasone, a halogen-containing, synthetic steroid, as 1°, 2°, or 3°. CH,OH CH3 OH HO. CH H CH3 ... dexamethasonearrow_forward18. Ketone reduction Dicyclohexyl ketone Reduce the ketone. 1. NaBH4, ethanol 2. H3O+ H OH Dicyclohexylmethanol (88%) (a 2° alcohol)arrow_forward
- Give the IUPAC name for each compound. CH3 CH2CH3 Br a. PHCH(CH3)2 b. С. d.arrow_forward1. Simple ketones, like acetone, are often used as industrial solvents for many organically based products such as adhesives and paints. They are considered "universal solvents," because they dissolve so many diverse materials. Explain why these chemicals are good solvents.arrow_forwardName each aldehyde or ketone. CH, CH, c. CH3-C-CH-CH-CH-C-H ČH,arrow_forward
- taken in order to gor the product Please explain the mechanisms/stepsarrow_forwardWhich of the following is phenol? CH2OH он I. IV. CH IL. V. OCH3 I. IV V.arrow_forwardMatch the letter corresponding to each structure to its proper functional group name. A. H,C CH В. H.C CH, C. HC-C-OH Ketone [ Choose ] [Choose] Alkene Carboxylic Acid Ketone Aldehydearrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

