
Concept explainers
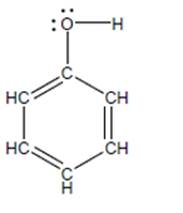
Phenol (shown below) has a
a. Based on
b. Draw the conjugate base of phenol (called phenoxide) including allimportant resonance structures.
c. Construct an explanation for why phenol is a stronger acid than anordinary alcohol. (You may want to consider first why phenoxide is lowerin PE than methoxide
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: A Guided Inquiry
- 5. sy For each reaction, identify the acid, base, conjugate acid, and conjugate base. Determine which direction the reaction will favor. Ka H₂SO3 = 1.54 x 10-2 and 1.07 x 10-7 Kb of CH3NH₂ = 4.4 x 10-4 Ka of CH3OH = 3.20 x 10-16 a. CH3O+ NH3 CH3OH + NH₂ b. HSO3 + CH3NH2 H₂SO3 + CH3NH3* c. 2HF + Ca(OH)2 2H₂O + CaF2arrow_forwardDraw the most likely conjugate base resulting from this acid-base reaction. Include all lone pairs. Ignore inorganic byproducts. NaOCH2CH3 Draw Conjugate Base N aarrow_forwardDraw the major organic product of the Bronsted acid-base reaction. Include all lone pairs and charges as appropriate. Ignore any counterions. H I N. NaOH H oarrow_forward
- Draw the products of each proton transfer reaction. Label the acid and base in the starting materials, and the conjugate and base in the products. а. СH,он + NH c. CH;CH2-0-H + H-Br b. CH;CH d. CH;C=C- + H2O + CH;O =arrow_forwardleft 4. Consider the following equilibrium: 29925610 + Aga weak and Sung base Bast CORS + 1ght base: Strong ad d AddOH ress stable OH Is the carboxylate anion (CH3CH₂COO) an appropriate base to deprotonate the alcohol? A) yes, because the equilibrium favors the left side B) yes, because the equilibrium favors the right side C) no, because the equilibrium favors the left side D) no, because the equilibrium favors the right side OH COO basarrow_forwardhydrogens on a carbon adjacent to a carbonyl group are far more acidic than those not adjacent to a carbonyl group. The anion derived from acetone, for example, is more stable than is the anion derived from ethane. Account for the greater stability of the anion from acetone. CH,ČCH, H CH,CH, H Acetone Ethane pK, 20.2 pK, 51arrow_forward
- 1. Consider the following unequal equilibrium. Which two species in this equilibrium represent the weaker acid and the weaker base? CH3CH3 + CH3NH 1 2 O 1 and 4 2 and 3 1 and 2 3 and 4 ? → CH3CH₂ 3 CH3NH₂ 4arrow_forward1. Rank each set of three alcohols in terms of increasing acidity, "1" being least acidic, and "3" being most acidic. goos CCI3 CI3C. Cl3 а. ОН ОН OH b. OH ОН ОН OH С. HOarrow_forwardWhich of the following is the strongest base? O a. Cl-CH2CH2NH2 Ob. Cl-CH2CH2CH2NH2 O c. Cl-aromatic ring-NH2 O d. Cl-CH2 -NH2 O e. C12-aromatic ring -NH2arrow_forward
- Select the stronger base and then draw its conjugate acid below. CICH₂CH₂O or CH3CH₂O- • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • You do not have to include lone pairs in your answer. Ⓡ OO. n[1] ? ChemDoodlearrow_forward2. Identify the most acidic compound by labeling it with "M" and the least acidic compound with an "L". Hint: On a separate sheet, draw each conjugate base and assess their stability. NH₂ A OH H₂SO4 3. Which ONE of the following compounds has the highest boiling point? + B CO₂H C HO O OH D Earrow_forwardRank the below compounds in order of increased acidity and explain your choice. HO. H3C || HO. HO.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
