
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 5, Problem 25CTQ
Interpretation Introduction
Interpretation: The base with lower potential energy in bases mentioned below should be determined.
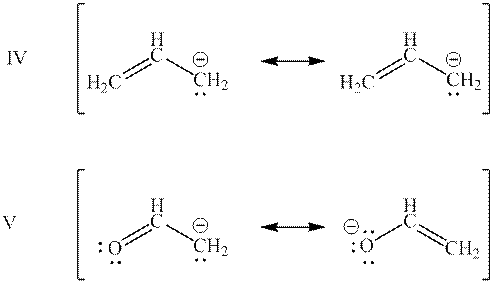
Concept introduction: When one single structure is unable to describe all the properties of single molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
✓
Check the box under each molecule that has a total of five ẞ hydrogens. If none of the molecules fit this description, check the box underneath the table.
tab
OH
CI
0
Br
xx
Br
None of these molecules have a total of five ẞ hydrogens.
esc
Explanation
Check
caps lock
shift
1
fn
control
02
F2
W
Q
A
N
#3
S
80
F3
E
$
t
01
205
%
5
F5
& 7
© 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility
FT
* 8
R
T
Y
U
כ
F6
9
FIG
F11
F
D
G
H
J
K
L
C
X
V B
<
N
M
H
option
command
P
H
+
F12
command
Draw the major product of this reaction. Ignore inorganic
byproducts and the carboxylic acid side product.
O
1. CHзMgBr (excess)
2. H₂O
✓
W
X
人
If cyclopentyl acetaldehyde reacts with NaOH, state the product (formula).
Chapter 5 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 5 - Which elements on the periodic table (other than...Ch. 5 - You will not find “hydroxide” in the stockroom,...Ch. 5 - Prob. 3CTQCh. 5 - Prob. 4CTQCh. 5 - Prob. 5CTQCh. 5 - Prob. 6CTQCh. 5 - On which do you expect to have a more intense and...Ch. 5 - Prob. 8CTQCh. 5 - Prob. 9CTQCh. 5 - Prob. 10CTQ
Ch. 5 - Prob. 11CTQCh. 5 - Prob. 12CTQCh. 5 - Prob. 13CTQCh. 5 - Prob. 14CTQCh. 5 - Prob. 15CTQCh. 5 - Prob. 16CTQCh. 5 - For each proposed set of resonance structures: a....Ch. 5 - Consider the polarization of the C=O bond in the...Ch. 5 - The C=O double bond is called a “carbonyl bond.”...Ch. 5 - Prob. 20CTQCh. 5 - Prob. 21CTQCh. 5 - Prob. 22CTQCh. 5 - Prob. 23CTQCh. 5 - Prob. 24CTQCh. 5 - Prob. 25CTQCh. 5 - Prob. 26CTQCh. 5 - Prob. 27CTQCh. 5 - Prob. 28CTQCh. 5 - Prob. 29CTQCh. 5 - Prob. 30CTQCh. 5 - Prob. 31CTQCh. 5 - Confirm that there is no legitimate Lewis...Ch. 5 - Draw all resonance structures of the molecule...Ch. 5 - Prob. 34CTQCh. 5 - Prob. 35CTQCh. 5 - Prob. 36CTQCh. 5 - Occasionally, we will see an ionic compound that...Ch. 5 - Prob. 2ECh. 5 - Prob. 3ECh. 5 - Prob. 4ECh. 5 - Is it possible to draw a resonance structure of...Ch. 5 - Prob. 6ECh. 5 - Prob. 7ECh. 5 - Prob. 8ECh. 5 - Phenol (shown below) has a pKa10 . a. Based on pKa...Ch. 5 - Use curved arrows to show the most likely...Ch. 5 - Prob. 12ECh. 5 - Complete each Lewis structure, draw all important...Ch. 5 - Use curved arrows to show the most likely...Ch. 5 - Construct an explanation for why sulfuric acid is...Ch. 5 - Prob. 16ECh. 5 - Prob. 17ECh. 5 - Prob. 18E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the major product of this reaction. Ignore inorganic byproducts. N S S HgCl2, H2SO4 く 8 W X Parrow_forwardtab esc く Drawing the After running various experiments, you determine that the mechanism for the following reaction occurs in a step-wise fashion. Br + OH + Using this information, draw the correct mechanism in the space below. 1 Explanation Check F2 F1 @2 Q W A os lock control option T S # 3 80 F3 Br $ 4 0105 % OH2 + Br Add/Remove step X C F5 F6 6 R E T Y 29 & 7 F D G H Click and drag to start drawing a structure. © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Ce A F7 DII F8 C Ո 8 * 9 4 F10 F C J K L C V Z X B N M H command P ge Coarrow_forwardIndicate compound A that must react with ethylbenzene to obtain 4-ethylbenzene-1-sulfonic acid. 3-bromo-4-ethylbenzene-1-sulfonic acid.arrow_forward
- Part 1 of 2 Draw the structure of A, the minor E1 product of the reaction. esc I Skip Part Check H₂O, D 2 A + Click and drag to start drawing a structure. -0- F1 F2 1 2 # 3 Q A 80 F3 W E S D F4 $ 4 % 5 F5 ㅇ F6 R T Y F G X 5 & 7 + Save 2025 McGraw Hill LLC. All Rights Reserved. DII F7 F8 H * C 80 J Z X C V B N 4 F9 6arrow_forwardFile Preview The following is a total synthesis of the pheromone of the western pine beetle. Such syntheses are interesting both because of the organic chemistry, and because of the possibility of using species specific insecticides, rather than broad band insecticides. Provide the reagents for each step. There is some chemistry from our most recent chapter in this synthesis, but other steps are review from earlier chapters. (8 points) COOEt COOEt A C COOEt COOEt COOH B OH OTS CN D E See the last homework set F for assistance on this one. H+, H₂O G OH OH The last step is just nucleophilic addition reactions, taking the ketone to an acetal, intramolecularly. But it is hard to visualize the three dimensional shape as it occurs. Frontalin, pheromone of the western pine beetlearrow_forwardFor the reaction below: 1. Draw all reasonable elimination products to the right of the arrow. 2. In the box below the reaction, redraw any product you expect to be a major product. C Major Product: Check + ◎ + X ง © Cl I F2 80 F3 I σ F4 I F5 NaOH Click and drawing F6 A 2025 McGraw Hill LLC. All Rights E F7 F8 $ # % & 2 3 4 5 6 7 8 Q W E R T Y U A S D F G H Jarrow_forward
- Can I please get help with this graph. If you can show exactly where it needs to pass through.arrow_forwardN Draw the major product of this reaction. Ignore inorganic byproducts. D 1. H₂O, pyridine 2. neutralizing work-up V P W X DE CO e C Larrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts. N O' 1. H2O, pyridine 2. neutralizing work-up く 8 W aarrow_forward
- Ideal Gas Law Practice Name If you need a refresher on Ideal Gas Law, go back to your Ideal Gas Law Reading Assignment from last week! On all of the following, you'll need to make sure to convert pressures to atm and convert temperatures to Kelvin in order to be able to use the R gas constant on your equation sheet! Given: Ideal Gas Law = then P= pressure V = volume R= ideal gas consent PV=nRT namount of substance n=PV/TR P=nRT/V I = temperature V=nRT/P T=PV/nR R=PV/nT 1. What pressure is required to contain 0.023 moles of nitrogen gas in a 4.2 L container at a temperature of 20.°C? 2. Oxygen gas is collected at a pressure of 123 kPa in a container which has a volume of 10.0 L. What temperature must be maintained on 0.500 moles of this gas in order to maintain this pressure? Express the temperature in degrees Celsius. 3. How many moles of chlorine gas would occupy a volume of 35.5 L at a pressure of 100.0 kPa and a temperature of 100. °C? After determining the number of moles,…arrow_forward1. The following conversion includes chemistry we have covered very recently, some chemistry from last term, and chemistry from the first chapter of this unit. Provide curly arrows and an explanation for this mechanism. Use the reagents in the order given. You do not need any other reagents. 1. NaOEt OEt 2.arrow_forwardCOOEt COOEt Step 1 Step 2 Step 3 COOEt COOEt COOH Step 6 OH Step 4 Step 7 (racemic) cyclizes under conditions (8) OTS Step 5 Step 8 ОН OH (racemic) Frontalin (racemic) Shown above are the steps in one of the several published syntheses of Frontalin, a pheromone of the western pine beetle. From the choices provided, show the reagents and conditions by which step 3 of this synthesis might be accomplished. List the reagent(s) in order that will accomplish this transformation. No more than 4 steps are required. List your answer as a single letter (single-step transformation) or a series of letters (multi-step transformation) with no commas separating them. For example, "ab" corresponds to: 1. Eto Na+ 2. NaOH, H₂O NOTE: The order in which you list your letters matters! Reagents: a. Eto Na* g. NaCN b. NaOH, H₂O h. SOCI₂ c. H3O+, heat i. (CH3)2CuLi, ether, -78°C d. LiAlH4 j. H₂O e. p-TsCI, pyridine k. RCO3H f. Br I. H3O+ 1,024arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning