
Concept explainers
(a)
Interpretation: Categorization of resonance structures as “Equivalent” and “Not equivalent” resonance structure for the bases mentioned below should be determined.
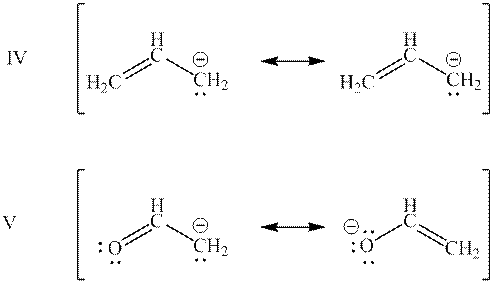
Concept introduction: When one single structure is unable to describe all the properties of single molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
(b)
Interpretation: Resonance structure in base mentioned below that has lowest potential energy should be determined.
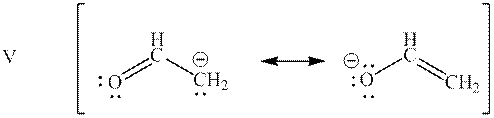
Concept introduction: When one single structure is unable to describe all the properties of single molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: A Guided Inquiry
- Be sure to answer all parts. The pk of three different C-H bonds is given below. For each compound, draw the conjugate base, including all possible resonance structures for compound C. Use skeletal structures when drawing all conjugate bases. compound A H H H pK₂ = 50 Conjugate base of compound A: Conjugate base of compound B: Conjugate base of compound C: edit structure ... compound B resonance structure (negative charge on C) H H edit structure ... (one Lewis structure) edit structure ... (one resonance structure) X H pka = 43 compound C edit structure ... H H pK₂ = 19.2 resonance structure (negative charge on O) Harrow_forwardO REPRESENTATIONS OF ORGANIC MOLECULES Ranking resonance structures Rank the resonance structures in each row of the table from major to minor. For example, in the first row, select (major) for the major resonance contributor. If two or more structures in the same row contribute equally, rank them equally by selecting the same number. H. H H. H. H. H. H. H. (Rank) (Rank) (Rank) H. H. H. H. H. H. H. (Rank) (Rank) :o: Rank) (Rank) 74°F DELL F5 F6 F7 F8 F9 F10 F11 PrtScr F12 24arrow_forwardHelp! a.) Draw all the important resonance contributors for the structure shown below. Use curved arrows to show the movement of electrons between these contributors. b.) Draw the hybrid resonance hybrid structure.arrow_forward
- A. For the molecule circled, draw the potential energy using this diagram. B. Draw the other three Newman projections based on the drawing at the bottom leftarrow_forwarda. Which of the following has a greater electronegativity? (circle one) Carbon / Cesium b. Explain why you chose your answer in Za.arrow_forwardRead This! The attractive and repulsive forces in an atom are rather complex. An electron is attracted to the protons in the nucleus, but it is also repelled by the other electrons in the atom. It is important to note however that the attractive force of the nucleus is NOT divided up among the electrons in the atom. Each electron gets approximately the full attractive force of the nucleus (minus the repulsive effects of other electrons). Compare the diagram below to set D in Model 3. Notice the similarity in attractive force. 0.10 nm 0.10 nm approx. 4.60 x 10-8 (on each electron) Model 4 – Period 3 Elements Aluminum Chlorine Sodium What does it MEAN? What do you WONDER? What do you SEE?arrow_forward
- ( do 4 with explanation )arrow_forward2. Each resonance structure of a given molecule contributes to the resonance hybrid. The more stable the resonance structure, the more that resonance structure contributes to the resonance hybrid. For the following resonance structures, a. Circle the resonance structure that contributes most to the resonance hybrid. b. Explain your choice for a in complete sentences. c. Draw the resonance hybrid. d. Add curved arrows to indicate the flow of electrons in each structure except the D. H A Q Search B hyji C Darrow_forward9. The two molecules shown below are structurally very similar. Draw all resonance structure for each species with curved arrows and circle which is molecule (A or B) is more stable because it has more resonance structures. Only use patterns 2 and 3. A Barrow_forward
- ( explain well with deeply explanation of each point of each question )arrow_forward(do last four asap )arrow_forward2. Tetrahydrocannabinolic acid (THCA), the precursor to THC, a psvchoactive: H.. • This molecule is missing eight lone pairs. Add them to the structure above • Check the formal charges of the oxygen atoms above. Do any of them have a non-zero formal charge? If so, add the charges to the appropriate oxygen atoms • THCA is a weak acid. Identify and circle the acidic hydrogen on the above structure • Draw a resonance structure of this molecule belowarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
