
Interpretation: A graph of a boiling point versus the number of Chlorine atoms for the five compounds listed in the Table 23.3 is to be drawn. Using the graph, the relationship between boiling point and degree of halogen substitution is to be described.
Concept Introduction: The boiling point is the temperature at which the liquid’s vapor pressure equalizes with the atmospheric pressure of the liquid’s environment.
Answer to Problem 64A
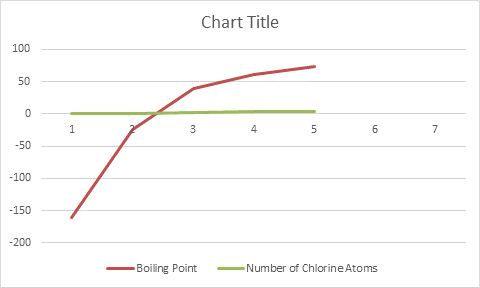
The graph is not a straight line. When hydrogen is replaced by halogen, the boiling point increases.
Explanation of Solution
Given Information: There is a relationship between boiling point and the degree of halogen substitution. Data in table 23.3 to draw a graph between boiling point and Chlorine atoms.
As given in table 23.3, the degree of halogen substitution increases from Methane Tetrachloromethane.
When hydrogen is replaced by a halogen, the boiling point increases due to an increase in the size of the halogen, London Dispersion Forces, and Dipole-Dipole attractions.
An increase in the degree of halogen substitution results in an increase in the surface area that leads to an increase in London Dispersion Forces, which then results in a higher boiling point.
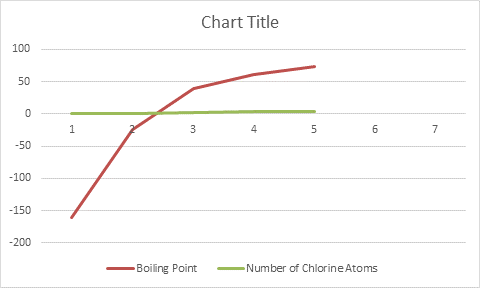
Graph is not a straight line. When hydrogen is replaced by halogen, the boiling point increases.
Chapter 23 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





