
a)
Interpretation: To draw the structural formula for the products of the reactions given.
Concept Introduction: Addition reactions are those types of reactions in which two or more molecules join together to form a larger molecule. These types of reactions will form additive products.
a)
Answer to Problem 52A
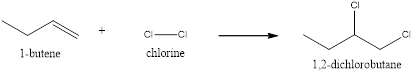
The name of the given structure is 1,2-dichlorobutane.
Explanation of Solution
In the given reaction, the double bond break and the bond between the chlorine molecule. The double bond of the butene breaks and the bond between the chlorine break generating the chloride atom. The chlorine will attack the butene which results in the formation of 1,2-dichlorobutane.
b)
Interpretation: To draw the structural formula for the products of the reactions given.
Concept Introduction: Addition reactions are those types of reactions in which two or more molecules join together to form a larger molecule. These types of reactions will form additive products.
b)
Answer to Problem 52A

Explanation of Solution
In the given reaction, the double bond break and the bond between the bromine molecule. The double bond of the butene breaks and the bond between the bromine break generate the bromide ion. The bromine will attack the butene which results in the formation of 1,2-dibromoobutane.
c)
Interpretation: To draw the structural formula for the products of the reactions given.
Concept Introduction: Addition reactions are those types of reactions in which two or more molecules join together to form a larger molecule. These types of reactions will form additive products.
c)
Answer to Problem 52A

Explanation of Solution
In this reaction, the HBr will add the following anti-Markovnikov’s rule. This reaction will take place in the presence of peroxide which results in the free radical mechanism and initiate the reaction and add bromine radical. This mechanism results in the product which is Bromo cyclohexane in this case.
Chapter 23 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





