
Interpretation : The boiling point of dodecane is to be predicted. The actual boiling point of dodecane is to be found. The actual boiling point with prediction is to be compared.
Concept Introduction : The temperature at which a liquid's vapor pressure equals the surrounding atmosphere is known as the boiling point of the liquid. This temperature causes the liquid to become a vapor.
Answer to Problem 84A
The predicted boiling point of dodecane is
The actual boiling point of dodecane is
The actual boiling point of dodecane differs from the predicted boiling point.
Explanation of Solution
The graph plotted between the number of carbons and temperature is as follows:
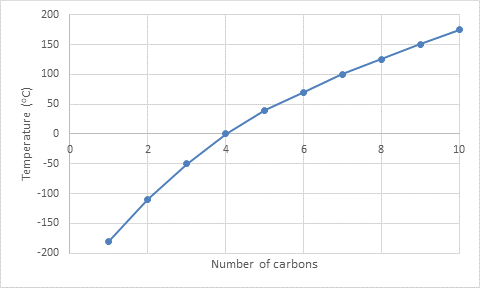
Dodecane is a straight-chain
But in the graph, there is plotting only 10 carbons.
To find the boiling point of dodecane from the graph, the graph has to be extrapolated.
According to the graph, the boiling point of dodecane is
But the actual boiling point of dodecane is
The actual boiling point differs from the predicted boiling point.
This is because the graph plotted is curved.
Since the predicted boiling point is an imaginary value.
Chapter 22 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





