
(a)
Interpretation- To name the given compound.
Concept Introduction-
- Identify the longest parent chain.
- Identify the substituents.
- Number each carbon in the parent chain in such a way that substituents get the lowest numbers.
- If the substituents are more than one, prefixes like di, tri, tetra etc are used.
- The name of compound starts with substituents with their position in parent chain. They are written in alphabetical order.
- Number and name are separated by dash while number are separated by commas.
- The chain name is based on the number of carbon atom present like meth, eth, prop, but, pent, hex, hept, oct, non, dec,…
- Suffix “ane” is used for
alkane parent chain and “yl is used for substituents.
(a)
Answer to Problem 45A
The name of compound is
Explanation of Solution
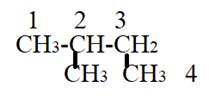
The compound has the longest parent chain with four carbon atoms. Hence the parent name chain will be ‘but’ with suffix “ane”.
Substituent has one carbon atom, and it is present on second carbon atom of the parent chain. Hence the name will be
Therefore, the name of compound is
Therefore, the name of compound is
(b)
Interpretation- To name the given compound.
Introduction- IUPAC nomenclature follows some rule for naming the compounds as:
- Identify the longest parent chain.
- Identify the substituents.
- Number each carbon in the parent chain in such a way that substituents get the lowest numbers.
- If the substituents are more than one, prefixes like di, tri, tetra etc are used.
- The name of compound starts with substituents with their position in parent chain. They are written in alphabetical order.
- Number and name are separated by dash while number are separated by commas.
- The chain name is based on the number of carbon atom present like meth, eth, prop, but, pent, hex, hept, oct, non, dec, …
Suffix “ane” is used for alkane parent chain and “yl is used for substituents
(b)
Answer to Problem 45A
The name of compound is
Explanation of Solution
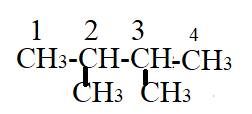
The compound has the longest parent chain with four carbon atoms. Hence the parent name chain will be “but” with suffix “ane”.
There are two methyl substituents present on second and third carbon atom of parent chain. Prefix “di” will be used. Hence the name will be
Therefore, the name of compound is
Therefore, the name of compound is
(c)
Interpretation- To name the given compound.
Introduction- IUPAC nomenclature follows some rule for naming the compounds as:
- Identify the longest parent chain.
- Identify the substituents.
- Number each carbon in the parent chain in such a way that substituents get the lowest numbers.
- If the substituents are more than one, prefixes like di, tri, tetra etc are used.
- The name of compound starts with substituents with their position in parent chain. They are written in alphabetical order.
- Number and name are separated by dash while number are separated by commas.
- The chain name is based on the number of carbon atom present like meth, eth, prop, but, pent, hex, hept, oct, non, dec,…
- Suffix “ane” is used for alkane parent chain and “yl is used for substituents.
(c)
Answer to Problem 45A
The name of compound is
Explanation of Solution
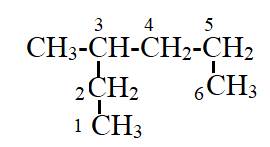
The compound has the longest parent chain with six carbon atoms. Hence the parent name chain will be hex with suffix “ane”.
Substituent has one carbon atom, and it is present on third carbon atom of the parent chain. Hence the name will be
Therefore, the name of compound is
Therefore, the name of compound is
Chapter 22 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





