
(a)
Interpretation − To draw the structural formula of given compound.
Introduction −
Structural formula is representation of molecule that shows arrangement of atoms and bonds.
Steps to draw the structural formula:
- Analyse the type of hydrocarbon using suffix in name.
- Prefixes indicate the type of substituents and the times of each substituent. The number indicate the position of substituent
- Start with drawing all the carbon -carbon bonds.
- Carbon hydrogen bonds are understood.
- For benzene, six carbon ring is drawn with alternate double bonds in it ..
(a)
Explanation of Solution
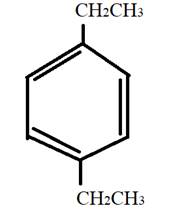
The given compound is
It has two same substituents ethyl at first and fourth carbon atom of benzene ring. Therefore, the structural formula can be:
(b)
Interpretation: To draw the structural formula of given compound.
Introduction −
Structural formula is representation of molecule that shows arrangement of atoms and bonds.
Steps to draw the structural formula:
- Analyse the type of hydrocarbon using suffix in name.
- Prefixes indicate the type of substituents and the times of each substituent. The number indicate the position of substituent
- Start with drawing all the carbon -carbon bonds.
- Carbon hydrogen bonds are understood.
(b)
Explanation of Solution
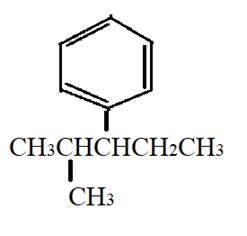
The given compound is
It has two substituents, methyl at second carbon atom and phenyl group at third carbon atom. It is an
Therefore, the structural formula can be:
(c)
Interpretation- To draw the structural formula of given compound.
Introduction −
Structural formula is representation of molecule that shows arrangement of atoms and bonds.
Steps to draw the structural formula:
- Analyse the type of hydrocarbon using suffix in name.
- Prefixes indicate the type of substituents and the times of each substituent. The number indicate the position of substituent
- Start with drawing all the carbon -carbon bonds.
- Carbon hydrogen bonds are understood.
- For benzene, six carbon ring is drawn with alternate double bonds in it ..
(c)
Explanation of Solution
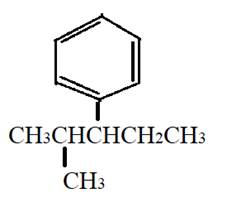
The given compound is
It has two same substituents, methyl at first and third carbon atom of benzene ring.
The structural formula can be:
Chapter 22 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





