
Concept explainers
(a)
Interpretation:
The keto-enol tautomer from the given pairs should be determined.
Concept Introduction:
Enol formation:
Under acidic conditions, alkyne reacts with water to produce an enol which then immediately converts into
The formed enol and ketone are called keto-enol tautomer.
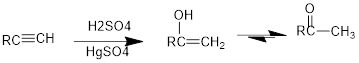
Tautomerization: It is the process of inter conversion of an enol compound to a ketone compound and therefore the process can also be called as keto-enol tautomerization.
(b)
Interpretation:
The keto-enol tautomers from the given pairs should be determined.
Concept Introduction:
Enol formation:
Under acidic conditions, alkyne reacts with water to produce an enol which then immediately converts into ketone. For terminal alkynes, there is a need of catalyst which is mercury.
The formed enol and ketone are called keto-enol tautomers.
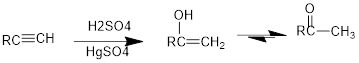
Tautomerization: It is the process of inter conversion of an enol compound to a ketone compound and therefore the process can also be called as keto-enol tautomerization.
(c)
Interpretation:
The keto-enol tautomers from the given pairs should be determined.
Concept Introduction:
Enol formation:
Under acidic conditions, alkyne reacts with water to produce an enol which then immediately converts into ketone. For terminal alkynes, there is a need of catalyst which is mercury.
The formed enol and ketone are called keto-enol tautomers.
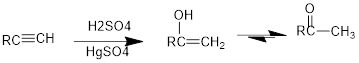
Tautomerization: It is the process of inter conversion of an enol compound to a ketone compound and therefore the process can also be called as keto-enol tautomerization.
(d)
Interpretation:
The keto-enol tautomers from the given pairs should be determined.
Concept Introduction:
Enol formation:
Under acidic conditions, alkyne reacts with water to produce an enol which then immediately converts into ketone. For terminal alkynes, there is a need of catalyst which is mercury.
The formed enol and ketone are called keto-enol tautomers.
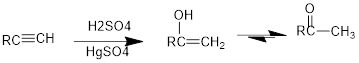
Tautomerization: It is the process of inter conversion of an enol compound to a ketone compound and therefore the process can also be called as keto-enol tautomerization.
(e)
Interpretation:
The keto-enol tautomers from the given pairs should be determined.
Concept Introduction:
Enol formation:
Under acidic conditions, alkyne reacts with water to produce an enol which then immediately converts into ketone. For terminal alkynes, there is a need of catalyst which is mercury.
The formed enol and ketone are called keto-enol tautomers.
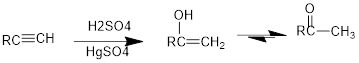
Tautomerization: It is the process of inter conversion of an enol compound to a ketone compound and therefore the process can also be called as keto-enol tautomerization.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Essential Organic Chemistry, Global Edition
- Draw the tautomer of this enol. Include all lone pairs. Ignore inorganic byproducts. :OH: H3O+ Draw Tautomerarrow_forwardOf the following structures, which is the most stable enol form?arrow_forwardTautomers are constitutional isomers that differ in the location of adouble bond and a hydrogen atom. Two tautomers are in equilibriumwith each other. Explain this ?arrow_forward
- OF OFFICI NSGR my Which of the following aldehydes would be the most soluble in water? (Hint: Remember the relation between the number of carbons in aldehydes and ketones and their solubility in water.) O CH3–CH2–CH2–CH2–CHO O CH3-CH2–CH2–CH2–CH2–CHO O CH3-CH2–CH2–CH2–CH2–CH2–CHO O CH3–CH2–CH2–CHO H f5 % 5 (O & hip 4+ CO fgarrow_forwardClassify each of the following transformations as either a substitution, elimination, addition orrearrangement reactionarrow_forward4) Give an example of an enol which would tautomerize into a) an aldehyde and b) a ketone.arrow_forward
- Draw the major organic substitution product for the reaction shown. он CH;CH2OH isopropyl alcohol Incorrectarrow_forwardDraw the tautomer of this ketone. Include all lone pairs. Ignore inorganic byproducts. :0: H3O+ 0 ✔arrow_forwardProvide the major organic product of the following reaction. but-1-yne 1) Sia₂BH 2) H₂O₂, OH What is it called when an enol converts to a keto? A) Keto-enol isomerism B) Keto-enol tater tot-ism C) Keto-enol conversion D) Keto-enol tautomerismarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





