
Interpretation:
The
Concept Introduction:
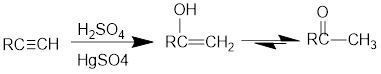
Acid Catalysed addition of water: When water is added to alkyne in the presence of an acid, the product formed will be an enol. Enol contains a double bond and
If a carbonyl group is bonded to two alkyl groups, it is called as a ketone. The enol formed in the acid catalysed addition of water will be easily converted into ketone.
Conversion of terminal
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Essential Organic Chemistry, Global Edition
- With which of the following can ethyl ethanoate not undergo a substitution reaction? * a-aqueous NaOH b-CH3OH, H+ c-aqueous NH3 d-CH3COONaarrow_forwardWhat are the major organic products obtained from the following reaction?arrow_forwardTrue or False Considering that two carbon chains have equal number of carbons, but one has Fluorine and the other has Iodine, the one with iodine will have a higher boiling point. Mild oxidation of alkenes results to similar product as that of nucleophilic addition of water to aldehydes.arrow_forward
- What explains why many aldehydes and ketones can undergo self- condensation reactions in basic conditions? The alpha carbon can lose a proton and act like a nucleophile and the carbonyl carbon a an electrophile O The alpha carbon can gain a proton and act like an electrophile and the carbonyl carbon is a nucleophile O The oxygen of the carbonyl group can attack the carbon of the carbonyl group Only esters can undergo self-condensation reactionsarrow_forwardWhat are the major organic products are formed when the following compounds react with methylmagnesium bromide (CH3MgBr), followed by the addition of dilute acid? propanal 2-pentanonearrow_forwardReaction with which of the following compounds in an acetylide reaction would lengthen the carbon chain by one carbon and add a primary alcohol? ethanal formaldehyde epoxide acetone carbonic acidarrow_forward
- What two alkenes give rise to each alcohol as the major product of acid-catalyzed hydration?arrow_forwardMatch the product in (Columns )when the following compounds in (Rows) are oxidized with K2Cr2O7? benzophenone benzoic acid no reaction benzaldehyde cyclohexanone What is the product produced when methanol reacts with benzoic acid? What is the name of the substance produced when methanol reacts with benzoic acid?arrow_forwardTo prepare an ether from an alkyl halide by a nucleophilic substitution reaction, what are the two types of nucleophiles that can be used?arrow_forward
- When trans-2-chloro-1-cyclohexanol is treated with a base, cyclohexene oxide is the product. However, when cis-2-chloro-1-cyclohexanol is treated with a base, the product is cyclohexanone, whyarrow_forwardWhat is the major organic product formed in the following reaction?arrow_forwardWhich of the following reactants would lead to an ester product as the major organic product? ? CH₂=CH2 CH3OH CH3CH3 CH3COOH + Esterarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,


