
Concept explainers
(a)
Interpretation:
The Lewis dot structure for TeCl2 must be drawn.
Concept Introduction :
Lewis dot structure is the representation of a molecule using valence electrons shown as dots.
Te is a p block element of group 16 with 6 valence electrons.
(a)
Answer to Problem 5E
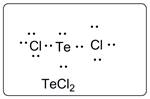
Lewis dot structure for TeCl2 is given below.
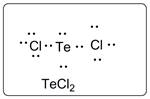
.
Explanation of Solution
In TeCl2, Te is the central atom which is bonded to two Cl atoms. There are three lone pairs of electrons on each Cl atom and two lone pair of electrons on Te atom.
Thus, the Lewis dot structure is as follows:
(b)
Interpretation:
The Lewis dot structure for HI must be drawn.
Concept Introduction :
Lewis dot structure is the representation of a molecule using valence electrons shown as dots.
I is a p block element of group 17 with 7 valence electrons.
(b)
Answer to Problem 5E
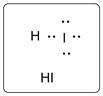
Lewis dot structure for HI is given below.
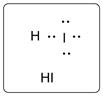
.
Explanation of Solution
In HI, I is bonded to one H atom. There are three lone pairs of electrons on I atom and hydrogen has zero lone pairs.
The structure is represented as follows:
(c)
Interpretation:
Lewis dot structures for AsBr3 must be drawn.
Concept Introduction :
Lewis dot structure is the representation of a molecule using valence electrons shown as dots.
As is a p block element of group 15 with 5 valence electrons.
(c)
Answer to Problem 5E
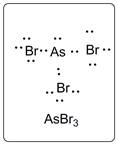
Lewis dot structure for AsBr3 is given below.
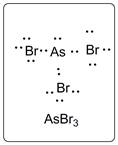
.
Explanation of Solution
In AsBr3, As is the central atom which is bonded to three Br atoms. There are one lone pair of electrons on As atom and three lone pairs of electrons on Br atom.
The structure is represented as follows:
(d)
Interpretation:
Lewis dot structures for SiF4 must be drawn.
Concept Introduction :
Lewis dot structure is the representation of a molecule using valence electrons shown as dots.
Si is a p block element of group 14 with 4 valence electrons.
(d)
Answer to Problem 5E
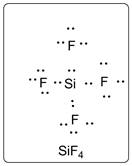
Lewis dot structure for SiF4 is given below.
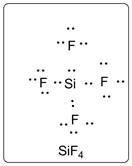
.
Explanation of Solution
In SiF4, Si is the central atom which is bonded to four F atoms. There are three lone pairs of electrons on F atom.
Structure is represented as follows:
(e)
Interpretation:
Lewis dot structures for F2 must be drawn.
Concept Introduction :
Lewis dot structure is the representation of a molecule using valence electrons shown as dots.
F is a p block element of group 17 with 7 valence electrons.
(e)
Answer to Problem 5E
Lewis dot structure for F2 is given below.
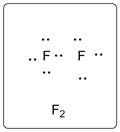
.
Explanation of Solution
In F2, both the F atoms are bonded to each other with a single bond. There are three lone pairs of electrons on each F atom.
The structure is represented as follows:
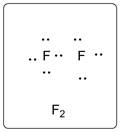
Chapter U2 Solutions
Living by Chemistry
Additional Science Textbook Solutions
Human Anatomy & Physiology (2nd Edition)
Campbell Essential Biology with Physiology (5th Edition)
Human Biology: Concepts and Current Issues (8th Edition)
Campbell Biology (11th Edition)
Microbiology: An Introduction
Biology: Life on Earth with Physiology (11th Edition)
- Indicate the correct option.a) Graphite conducts electricity, being an isotropic materialb) Graphite is not a conductor of electricityc) Both are falsearrow_forward(f) SO: Best Lewis Structure 3 e group geometry:_ shape/molecular geometry:, (g) CF2CF2 Best Lewis Structure polarity: e group arrangement:_ shape/molecular geometry: (h) (NH4)2SO4 Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forward1. Problem Set 3b Chem 141 For each of the following compounds draw the BEST Lewis Structure then sketch the molecule (showing bond angles). Identify (i) electron group geometry (ii) shape around EACH central atom (iii) whether the molecule is polar or non-polar (iv) (a) SeF4 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: (b) AsOBr3 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles):arrow_forward
- (c) SOCI Best Lewis Structure 2 e group arrangement: shape/molecular geometry:_ (d) PCls Best Lewis Structure polarity: e group geometry:_ shape/molecular geometry:_ (e) Ba(BrO2): Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forwardDon't used Ai solutionarrow_forwardDon't used Ai solutionarrow_forward
- reaction scheme for C39H4202 Hydrogenation of Alkyne (Alkyne to Alkene) show reaction (drawing) pleasearrow_forwardGive detailed mechanism Solution with explanation needed. Don't give Ai generated solutionarrow_forwardShow work with explanation needed....don't give Ai generated solutionarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





