
Interpretation: To show the reaction when internal neutralization takes place in the glycine molecule.
Concept introduction: Amino acids are amphoteric in nature since it has both carboxyl and amino acid groups which means they can behave as both acid and base.
Answer to Problem 10STP
The internal neutralization reaction of glycine can be depicted as follows:
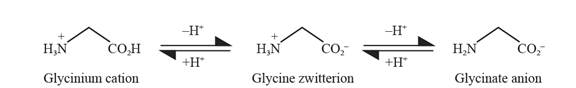
Explanation of Solution
Glycine can act as both acids as well bases. About all amino acids exist in zwitter ions.
The formation of zwitter ion in the case of glycine takes place as follows:
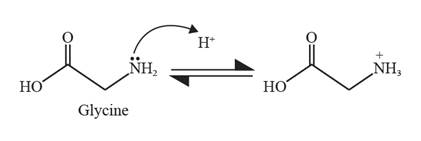
In strongly alkaline conditions, glycine can act as a base
The reaction of glycine with a base leads to the formation of a negative ion when an ammonium ion loses a proton.
The chemical equation can be depicted as follows:
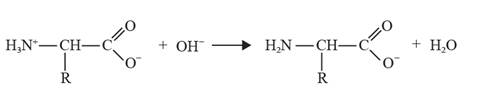
In acidic conditions, glycine can act as a base.
The reaction of glycine with acid leads to the formation of a positive ion as the carboxylate ion gains a proton.
The chemical equation can be depicted as follows:
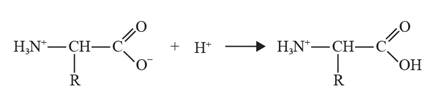
Yes, glycine can undergo an internal neutralization reaction and it forms a zwitter ion.
Chapter 24 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





