
Interpretation: The reaction between the given compound when hydrolyzed with boiling sodium hydroxide needs to be interpreted and the products of saponification to be given.
Concept introduction: A saponification reaction is one that involves the conversion of fat into soap and also the conversion of an alkali-metal hydroxide into alcohol.
The
Answer to Problem 109A
The products of saponification are glycerol and sodium salts i.e., soap molecules.
The chemical reaction showing saponification of the given compound is depicted as follows:
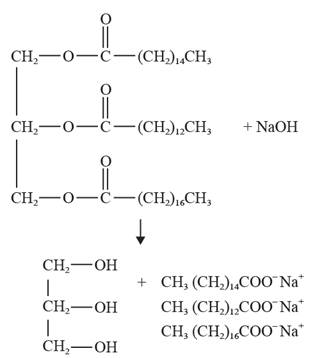
Explanation of Solution
The process which involves the hydrolysis of fats and oils in the presence of an aqueous solution of alkali-metal hydroxide is known as saponification. This process is beneficial in the formation of soaps which are salts (alkali-metal) of fatty acids.
The chemical reaction showing saponification of the given compound (fat) is as follows:
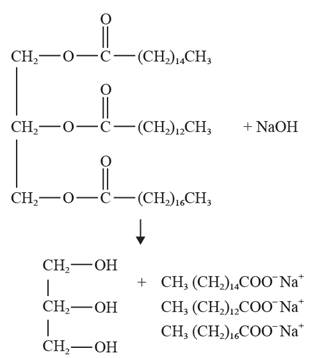
Thus, the products are glycerol and sodium salts i.e., soap molecules.
The products of saponification are glycerol and sodium salts i.e., soap molecules.
Chapter 24 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





