
(a)
Interpretation:
Whether the given compound is water soluble or insoluble needs to be determined.
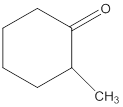
Concept Introduction:
The main rule which is used to determine the solubility of a compound in a given solution is that "like dissolves like". Therefore, the polar compounds will be soluble in polar solvents while the non-polar compounds will be soluble in non-polar solvents.
(b)
Interpretation:
Whether the given compound is water soluble or insoluble needs to be determined.
Concept Introduction:
The main rule which is used to determine the solubility of a compound in a given solution is that "like dissolves like". Therefore, the polar compounds will be soluble in polar solvents while the non-polar compounds will be soluble in non-polar solvents.
(c)
Interpretation:
Whether the given compound is water soluble or insoluble needs to be determined.
Concept Introduction:
The main rule which is used to determine the solubility of a compound in a given solution is that "like dissolves like". Therefore, the polar compounds will be soluble in polar solvents while the non-polar compounds will be soluble in non-polar solvents.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
- what is/are the major products) of this dehydration reaction? OH H-SO heat +. A. +. B. C. D.arrow_forwardMatch the given compounds with their expected solubility. soluble in 5% HCl soluble in 5% NaOH insoluble in water and concentrated H2SO4 soluble in water a. hexane b. decanoic acid c. methanol d. octanaminearrow_forwardWhich element is present in all organic compounds?A. Helium C. CarbonB. Nitrogen D. Oxygen 2. What products are obtained with CH 4 (g) burns completely in an excess ofoxygen? A. CO 2 C. Hydrogen gasB. CO 2 and H 2 O D. Explosion 3. A hydrocarbon molecule is saturated if the molecule contains ___________A. Single covalent bonds, only C. A double covalent bond, onlyB. A triple covalent bond D. Single and double covalent bonds4. A functional group with a carbonyl group functionality is _________________. A. Alcohol C. EsterB. Amine D. Ketone5. Choose the incorrect option regarding Isomerism: A. They differ in both physical and chemical propertiesB. They have the different molecular formulaC. Chain isomers differ in the arrangement of their skeletonD. They have the same molecular formula 6. Which among the following is formed when an alcohol is dehydrated? A. Aldehyde C. AlkeneB. Ketone D. Amine 7. The reaction of water with alkene to produce an alcohol is a/an ______reaction. A. Addition…arrow_forward
- 2. What type of chemical reaction is illustrated by the following reaction? CH3OOCH3 + H2O ⎯→ CH3OOH + CH3OH a. oxidation/reduction b. substitution c. neutralization d. hydrolysis e. dehydration synthesis (condensation)arrow_forwardwhich organic compound dissolves in water? 1. 2-pentanol 2. 1-hexanol 3. diethylether 4. 3-methyl-3-pentanol which organic compound dissolves in 10% NaOH? 1. 2-pentanol 2. 1-hexanol 3. diethylether 4. 3-methyl-3-pentanolarrow_forwardConsider the structure of Vitamin A1 (C20H300). Vitamin A1 should be CH3 CH3 CH3 CH2OH H2C H2Č. C' H2 C. CH3 CH3 Periodic Table and Datasheet Select one: Oa. insoluble in water and in oil O b. less soluble in water than in oil O c. equally soluble in water and in oil O d. more soluble in water than in oil CH CH HC CH CH HCarrow_forward
- 5. Draw a structural example of a primary, secondary and tertiary alcohol. 6. Classify the following reaction as oxidation, dehydration or hydration. a. CH.CH(OH)CH, CH,COCH, 7. Identify each compound as an alcohol or an ether. Classify any alcohols as primary, secondary or tertiary. он b. он HO d. 8. Write the product(s) for the following reactions. a. H,O b. H,Oarrow_forwardOH HO 7. OH The above is an example of a a. primary alcohol b. secondary alcohol c. tertiary alcohol Od. phenolarrow_forward3. Which is more effective as a disinfectant: ethyl alcohol or isopropyl alcohol? Choose only one and explain why. 4. What is the difference between cleaning, sanitizing, and disinfecting?arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

