
Concept explainers
Which compound in each pair has the higher boiling point?
a.
b. 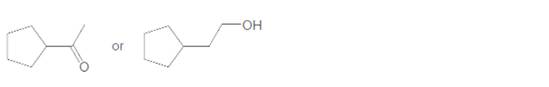
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
- explain why methanethiol, CH3SH, has a lower boiling point (6°C) than methanol, CH3OH (65°C), even though methanethiol has a higher molecular weightarrow_forwardDraw the structure of a molecule that fi ts each description: a. a 2 ° alcohol of molecular formula C 6H 12O b. a cyclic ether with molecular formula C 5H 10O c. a 1 ° alkyl halide with molecular formula C 5H 11Clarrow_forwardName each compound in which the benzene ring is best treated as a substituent. CH3 a. CH3-CH-CH,-CH-CH,-CH–CH,-CH; CH,-CH3 b. CH,-CH-CH=CH-CH,-CH,–CH,-CH, c. CH3-C=C-CH-CH-CH-CH2-CH3 CH3 CH3arrow_forward
- #544 of Isomers (chiral) Which of the following 2-chloro-1-butanol (A), 3-chloro-1-butanol (B), and 4-chloro-1-butanol (C), contain a stereogenic carbon atom. Select one: a. A only O b. B only O c. C only O d. A and B e. A, B and Carrow_forwardArrange the compounds in order of increasing boiling point. HO. B C D Aarrow_forwardClassify each alcohol as to 1º, 2º, or 3º 1. 2. CH3CH2CH(CH3)CH2OHCH3 3. CH2CH2CHOHCH3arrow_forward
- Select the compound with the highest boiling point at standard pressure. O a) O b) O c) o d) e) CH 3 CH 2CH 3 HOCH₂CH₂OH CH4 H3C-CH-CH3 OH CH₂ CH₂OH 3 2arrow_forwardArrange these compounds in order of increasing boiling point (values in C are 42, 24, 78, and 118). (a) CH3CH2OH (b) CH3OCH3 (c) CH3CH2CH3 (d) CH3COOHarrow_forwardWhich alcohol is the most soluble in water? A CH;CH,CHOHCH3 B CH;CH2CH2CH2OH (c) (CH3)2CHCH2OH D (CH3)3COHarrow_forward
- Give the IUPAC name for each compound. CH3 CH2CH3 Br a. PHCH(CH3)2 b. С. d.arrow_forward18. Ketone reduction Dicyclohexyl ketone Reduce the ketone. 1. NaBH4, ethanol 2. H3O+ H OH Dicyclohexylmethanol (88%) (a 2° alcohol)arrow_forwardDraw the products of combustion of each alkane. a. CH;CH,CH,CH2CH(CH3)2 b.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

